Distinct properties of stimulus-evoked bursts in the lateral geniculate nucleus
- PMID: 15647497
- PMCID: PMC6725468
- DOI: 10.1523/JNEUROSCI.3369-04.2005
Distinct properties of stimulus-evoked bursts in the lateral geniculate nucleus
Abstract
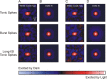
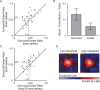
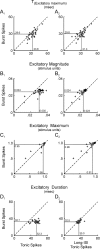
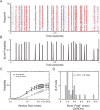
Neurons in the lateral geniculate nucleus (LGN) of the thalamus produce spikes that can be classified as burst spikes and tonic spikes. Although burst spikes are generally associated with states of sleep and drowsiness, bursts may also play an important role in sensory processing. This study explores the stimulus properties that evoke burst and tonic spikes and examines the reliability of LGN neurons to produce visually driven bursts. Using reverse-correlation techniques, we show that the receptive fields of burst spikes are similar to, but significantly different from, the receptive fields of tonic spikes. Compared with tonic spikes, burst spikes (1) occur with a shorter latency between stimulus and response, (2) have a greater dependence on stimuli with transitions from suppressive to preferred states, and (3) prefer stimuli that provide increased drive to the receptive field center and even greater increased drive to the receptive field surround. These differences are not attributable to the long interspike interval that precedes burst spikes, because tonic spikes with similar preceding interspike intervals also differ from burst spikes in both the spatial and temporal domains. Finally, measures of reliability are significantly greater for burst spikes than for tonic spikes with similar preceding interspike intervals. These results demonstrate that thalamic bursts contribute to sensory processing and can reliably provide the cortex with information that is similar to, but distinct from, that of tonic spikes.
Figures


References
-
- Albrecht DG (1995) Visual cortex neurons in monkey and cat: effect of contrast on the spatial and temporal phase transfer functions. Vis Neurosci 12: 1191-1210. - PubMed
-
- Benardete EA, Kaplan E, Knight BW (1992) Contrast gain control in the primate retina: P cells are not X-like, some M cells are. Vis Neurosci 8: 483-486. - PubMed
-
- Bishop PO, Burke W, Davis R (1958) Synapse discharge by single fibre in mammalian visual system. Nature 128: 728-730. - PubMed
-
- Carandini M, Heeger DJ (1994) Summation and division by neurons in primate visual cortex. Science 264: 1333-1336. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
