Resistance to Apo2 ligand (Apo2L)/tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-mediated apoptosis and constitutive expression of Apo2L/TRAIL in human T-cell leukemia virus type 1-infected T-cell lines
- PMID: 15650163
- PMCID: PMC544134
- DOI: 10.1128/JVI.79.3.1367-1378.2005
Resistance to Apo2 ligand (Apo2L)/tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-mediated apoptosis and constitutive expression of Apo2L/TRAIL in human T-cell leukemia virus type 1-infected T-cell lines
Retraction in
-
Retraction. Resistance to Apo2 ligand (Apo2L)/tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-mediated apoptosis and constitutive expression of Apo2L/TRAIL in human T-cell leukemia virus type 1-infected T-cell lines. Journal of Virology, Volume 79, no. 3, p. 1367-78, 2005.J Virol. 2011 Feb;85(3):1416. doi: 10.1128/JVI.02070-10. J Virol. 2011. PMID: 21228250 Free PMC article. No abstract available.
Abstract
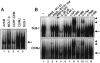
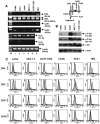
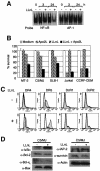
Adult T-cell leukemia (ATL), a CD4+-T-cell malignancy caused by human T-cell leukemia virus type 1 (HTLV-1), is difficult to cure, and novel treatments are urgently needed. Apo2 ligand (Apo2L; also tumor necrosis factor-related apoptosis-inducing ligand [TRAIL]) has been implicated in antitumor therapy. We found that HTLV-1-infected T-cell lines and primary ATL cells were more resistant to Apo2L-induced apoptosis than uninfected cells. Interestingly, HTLV-1-infected T-cell lines and primary ATL cells constitutively expressed Apo2L mRNA. Inducible expression of the viral oncoprotein Tax in a T-cell line up-regulated Apo2L mRNA. Analysis of the Apo2L promoter revealed that this gene is activated by Tax via the activation of NF-kappaB. The sensitivity to Apo2L was not correlated with expression levels of Apo2L receptors, intracellular regulators of apoptosis (FLICE-inhibitory protein and active Akt). NF-kappaB plays a crucial role in the pathogenesis and survival of ATL cells. The resistance to Apo2L-induced apoptosis was reversed by N-acetyl-L-leucinyl-L-leucinyl-lLnorleucinal (LLnL), an NF-kappaB inhibitor. LLnL significantly induced the Apo2L receptors DR4 and DR5. Our results suggest that the constitutive activation of NF-kappaB is essential for Apo2L gene induction and protection against Apo2L-induced apoptosis and that suppression of NF-kappaB may be a useful adjunct in clinical use of Apo2L against ATL.
Figures




Similar articles
-
Apo2 ligand/TNF-related apoptosis-inducing ligand and death receptor 5 mediate the apoptotic signaling induced by ionizing radiation in leukemic cells.Cancer Res. 2000 Oct 15;60(20):5754-60. Cancer Res. 2000. PMID: 11059770
-
Human T-cell leukemia virus type-I oncoprotein Tax inhibits Fas-mediated apoptosis by inducing cellular FLIP through activation of NF-kappaB.Genes Cells. 2006 Feb;11(2):177-91. doi: 10.1111/j.1365-2443.2006.00927.x. Genes Cells. 2006. PMID: 16436054
-
TRAIL/Apo2L ligands induce apoptosis in malignant rhabdoid tumor cell lines.Pediatr Res. 2003 Nov;54(5):709-17. doi: 10.1203/01.PDR.0000085038.53151.D0. Epub 2003 Aug 6. Pediatr Res. 2003. PMID: 12904602
-
NF-kappaB in pathogenesis and treatment of adult T-cell leukemia/lymphoma.Int Rev Immunol. 2007 Sep-Dec;26(5-6):269-81. doi: 10.1080/08830180701703364. Int Rev Immunol. 2007. PMID: 18027201 Review.
-
The multifaceted oncoprotein Tax: subcellular localization, posttranslational modifications, and NF-κB activation.Adv Cancer Res. 2012;113:85-120. doi: 10.1016/B978-0-12-394280-7.00003-8. Adv Cancer Res. 2012. PMID: 22429853 Review.
Cited by
-
Apoptotic killing of HIV-1-infected macrophages is subverted by the viral envelope glycoprotein.PLoS Pathog. 2007 Sep 7;3(9):1281-90. doi: 10.1371/journal.ppat.0030134. PLoS Pathog. 2007. PMID: 17907802 Free PMC article.
-
Human T-cell leukemia virus type 1 Tax-deregulated autophagy pathway and c-FLIP expression contribute to resistance against death receptor-mediated apoptosis.J Virol. 2014 Mar;88(5):2786-98. doi: 10.1128/JVI.03025-13. Epub 2013 Dec 18. J Virol. 2014. PMID: 24352466 Free PMC article.
-
Caspase control: protagonists of cancer cell apoptosis.Exp Oncol. 2012 Oct;34(3):165-75. Exp Oncol. 2012. PMID: 23070001 Free PMC article. Review.
-
Nef Is Dispensable for Resistance of Simian Immunodeficiency Virus-Infected Macrophages to CD8+ T Cell Killing.J Virol. 2015 Oct;89(20):10625-36. doi: 10.1128/JVI.01699-15. Epub 2015 Aug 12. J Virol. 2015. PMID: 26269172 Free PMC article.
-
Identification of bovine leukemia virus tax function associated with host cell transcription, signaling, stress response and immune response pathway by microarray-based gene expression analysis.BMC Genomics. 2012 Mar 28;13:121. doi: 10.1186/1471-2164-13-121. BMC Genomics. 2012. PMID: 22455445 Free PMC article.
References
-
- Akagi, T., H. Ono, and K. Shimotohno. 1995. Characterization of T cells immortalized by Tax1 of human T-cell leukemia virus type 1. Blood 86:4243-4249. - PubMed
-
- Almasan, A., and A. Ashkenazi. 2003. Apo2L/TRAIL: apoptosis signaling, biology, and potential for cancer therapy. Cytokine Growth Factor Rev. 14:337-348. - PubMed
-
- Ashkenazi, A., R. C. Pai, S. Fong, S. Leung, D. A. Lawrence, S. A. Marsters, C. Blackie, L. Chang, A. E. McMurtrey, A. Hebert, L. DeForge, I. L. Koumenis, D. Lewis, L. Harris, J. Bussiere, H. Koeppen, Z. Shahrokh, and R. H. Schwall. 1999. Safety and antitumor activity of recombinant soluble Apo2 ligand. J. Clin. Investig. 104:155-162. - PMC - PubMed
-
- Baetu, T. M., H. Kwon, S. Sharma, N. Grandvaux, and J. Hiscott. 2001. Disruption of NF-κB signaling reveals a novel role for NF-κB in the regulation of TNF-related apoptosis-inducing ligand expression. J. Immunol. 167:3164-3173. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

