Herpes simplex virus type 1 infection via the bloodstream with apolipoprotein E dependence in the gonads is influenced by gender
- PMID: 15650186
- PMCID: PMC544102
- DOI: 10.1128/JVI.79.3.1605-1612.2005
Herpes simplex virus type 1 infection via the bloodstream with apolipoprotein E dependence in the gonads is influenced by gender
Abstract
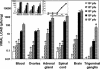
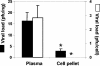
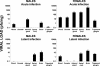
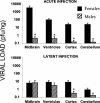
Herpes simplex virus type 1 (HSV-1) causes disease in humans and animals. Infection usually occurs via the neural route and possibly occurs via the hematogenous route. The latter, however, is the main route by which immunosuppressed individuals and neonates are infected. Gender-dependent differences in the incidence and severity of some viral infections have been reported. To detect differences between the sexes with respect to HSV-1 colonization and disease, the characteristics of both acute and latent infections in hematogenously infected male and female mice were compared. In acute infection, the female mice had a poorer outcome: HSV-1 colonization was more effective, especially in the gonads and brain. In the encephalon, the midbrain had the highest viral load. In latent infection, brain viral loads were not significantly different with respect to sex. Significant differences were seen, however, in the blood and trigeminal ganglia: HSV-1 seroprevalence was observed in females, with no virus detected in males. In brain dissections, only the cerebral cortex of the females had viral loads statistically higher than those observed in the males. The spread of the virus to several organs of interest during acute infection was examined immunohistochemically. Female mice showed greater viral immunostaining, especially in the adrenal cortex, gonads, and midbrain. In male mice, HSV-1 was detected predominantly in the adrenal cortex. It was also found that apolipoprotein E promotes virus colonization of the ovaries, the APOE gene dose being directly related to viral invasiveness.
Figures

Similar articles
-
Herpes simplex virus type 1 latency in the murine nervous system is associated with oxidative damage to neurons.Virology. 2000 Dec 20;278(2):309-21. doi: 10.1006/viro.2000.0678. Virology. 2000. PMID: 11118355
-
Factors affecting herpes simplex virus reactivation from the explanted mouse brain.Virology. 2012 Nov 10;433(1):116-23. doi: 10.1016/j.virol.2012.07.018. Epub 2012 Aug 11. Virology. 2012. PMID: 22884293
-
Establishment of latent herpes simplex virus type 1 infection in resistant, sensitive, and immunodeficient mouse strains.Virology. 2000 Mar 1;268(1):17-28. doi: 10.1006/viro.1999.0158. Virology. 2000. PMID: 10683323
-
The latency-associated gene of herpes simplex virus type 1 (HSV-1) interferes with superinfection by HSV-1.J Neurovirol. 2002 Dec;8 Suppl 2:97-102. doi: 10.1080/13550280290167920. J Neurovirol. 2002. PMID: 12491159 Review.
-
A role for apoptosis induced by acute herpes simplex virus infection in mice.Int Rev Immunol. 2004 Jan-Apr;23(1-2):173-85. doi: 10.1080/08830180490265583. Int Rev Immunol. 2004. PMID: 14690859 Review.
Cited by
-
Sex influences immune responses to viruses, and efficacy of prophylaxis and treatments for viral diseases.Bioessays. 2012 Dec;34(12):1050-9. doi: 10.1002/bies.201200099. Epub 2012 Sep 26. Bioessays. 2012. PMID: 23012250 Free PMC article.
-
Cell-mediated immunity to HIV in the female reproductive tract.J Reprod Immunol. 2009 Dec;83(1-2):190-5. doi: 10.1016/j.jri.2009.07.012. Epub 2009 Oct 25. J Reprod Immunol. 2009. PMID: 19857902 Free PMC article. Review.
-
Systemic inflammation as a central player in the initiation and development of Alzheimer's disease.Immun Ageing. 2025 Aug 21;22(1):33. doi: 10.1186/s12979-025-00529-5. Immun Ageing. 2025. PMID: 40841660 Free PMC article. Review.
-
Effect of apolipoprotein E on the cerebral load of latent herpes simplex virus type 1 DNA.J Virol. 2006 Jun;80(11):5383-7. doi: 10.1128/JVI.00006-06. J Virol. 2006. PMID: 16699018 Free PMC article.
-
Herpes Simplex Virus Type 1 Infection of the Central Nervous System: Insights Into Proposed Interrelationships With Neurodegenerative Disorders.Front Cell Neurosci. 2019 Feb 26;13:46. doi: 10.3389/fncel.2019.00046. eCollection 2019. Front Cell Neurosci. 2019. PMID: 30863282 Free PMC article. Review.
References
-
- Barna, M., T. Komatsu, Z. Bi, and C. S. Reiss. 1996. Sex differences in susceptibility to viral infection of the central nervous system. J. Neuroimmunol. 67:31-39. - PubMed
-
- Buijs, R. M., J. Wortel, J. J. Van Heerikhuize, M. G. Feenstra, G. J. Ter Horst, H. J. Romijn, and A. Kalsbeek. 1999. Anatomical and functional demonstration of a multisynaptic suprachiasmatic nucleus adrenal (cortex) pathway. Eur. J. Neurosci. 11:1535-1544. - PubMed
-
- Burgos, J. S., C. Ramirez, I. Sastre, M. J. Bullido, and F. Valdivieso. 2003. ApoE4 is more efficient than E3 in brain access by herpes simplex virus type 1. Neuroreport 14:1825-1827. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

