Inhibition of interferon signaling by the New York 99 strain and Kunjin subtype of West Nile virus involves blockage of STAT1 and STAT2 activation by nonstructural proteins
- PMID: 15650219
- PMCID: PMC544092
- DOI: 10.1128/JVI.79.3.1934-1942.2005
Inhibition of interferon signaling by the New York 99 strain and Kunjin subtype of West Nile virus involves blockage of STAT1 and STAT2 activation by nonstructural proteins
Abstract
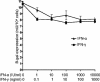
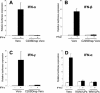
The interferon (IFN) response is the first line of defense against viral infections, and the majority of viruses have developed different strategies to counteract IFN responses in order to ensure their survival in an infected host. In this study, the abilities to inhibit IFN signaling of two closely related West Nile viruses, the New York 99 strain (NY99) and Kunjin virus (KUN), strain MRM61C, were analyzed using reporter plasmid assays, as well as immunofluorescence and Western blot analyses. We have demonstrated that infections with both NY99 and KUN, as well as transient or stable transfections with their replicon RNAs, inhibited the signaling of both alpha/beta IFN (IFN-alpha/beta) and gamma IFN (IFN-gamma) by blocking the phosphorylation of STAT1 and its translocation to the nucleus. In addition, the phosphorylation of STAT2 and its translocation to the nucleus were also blocked by KUN, NY99, and their replicons in response to treatment with IFN-alpha. IFN-alpha signaling and STAT2 translocation to the nucleus was inhibited when the KUN nonstructural proteins NS2A, NS2B, NS3, NS4A, and NS4B, but not NS1 and NS5, were expressed individually from the pcDNA3 vector. The results clearly demonstrate that both NY99 and KUN inhibit IFN signaling by preventing STAT1 and STAT2 phosphorylation and identify nonstructural proteins responsible for this inhibition.
Figures


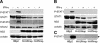
Similar articles
-
A single amino acid substitution in the West Nile virus nonstructural protein NS2A disables its ability to inhibit alpha/beta interferon induction and attenuates virus virulence in mice.J Virol. 2006 Mar;80(5):2396-404. doi: 10.1128/JVI.80.5.2396-2404.2006. J Virol. 2006. PMID: 16474146 Free PMC article.
-
Analysis of adaptive mutations in Kunjin virus replicon RNA reveals a novel role for the flavivirus nonstructural protein NS2A in inhibition of beta interferon promoter-driven transcription.J Virol. 2004 Nov;78(22):12225-35. doi: 10.1128/JVI.78.22.12225-12235.2004. J Virol. 2004. PMID: 15507609 Free PMC article.
-
Paramyxoviridae use distinct virus-specific mechanisms to circumvent the interferon response.Virology. 2000 Apr 10;269(2):383-90. doi: 10.1006/viro.2000.0240. Virology. 2000. PMID: 10753717
-
Dengue Virus Control of Type I IFN Responses: A History of Manipulation and Control.J Interferon Cytokine Res. 2015 Jun;35(6):421-30. doi: 10.1089/jir.2014.0129. Epub 2015 Jan 28. J Interferon Cytokine Res. 2015. PMID: 25629430 Free PMC article. Review.
-
Kunjin RNA replication and applications of Kunjin replicons.Adv Virus Res. 2003;59:99-140. doi: 10.1016/s0065-3527(03)59004-2. Adv Virus Res. 2003. PMID: 14696328 Review.
Cited by
-
Reversion to ancestral Zika virus NS1 residues increases competence of Aedes albopictus.PLoS Pathog. 2020 Oct 14;16(10):e1008951. doi: 10.1371/journal.ppat.1008951. eCollection 2020 Oct. PLoS Pathog. 2020. PMID: 33052957 Free PMC article.
-
Zika virus noncoding RNA cooperates with the viral protein NS5 to inhibit STAT1 phosphorylation and facilitate viral pathogenesis.Sci Adv. 2022 Dec 2;8(48):eadd8095. doi: 10.1126/sciadv.add8095. Epub 2022 Nov 30. Sci Adv. 2022. PMID: 36449607 Free PMC article.
-
The global ecology and epidemiology of West Nile virus.Biomed Res Int. 2015;2015:376230. doi: 10.1155/2015/376230. Epub 2015 Mar 19. Biomed Res Int. 2015. PMID: 25866777 Free PMC article. Review.
-
Inhibition of dengue virus polymerase by blocking of the RNA tunnel.J Virol. 2010 Jun;84(11):5678-86. doi: 10.1128/JVI.02451-09. Epub 2010 Mar 17. J Virol. 2010. PMID: 20237086 Free PMC article.
-
Differential expression of interferon (IFN) regulatory factors and IFN-stimulated genes at early times after West Nile virus infection of mouse embryo fibroblasts.J Virol. 2007 Nov;81(21):12005-18. doi: 10.1128/JVI.01359-07. Epub 2007 Sep 5. J Virol. 2007. PMID: 17804507 Free PMC article.
References
-
- Diamond, M. S. 2003. Evasion of innate and adaptive immunity by flaviviruses. Immunol. Cell Biol. 81:196-206. - PubMed
-
- Foy, E., K. Li, C. Wang, R. Sumpter, Jr., M. Ikeda, S. M. Lemon, and M. Gale, Jr. 2003. Regulation of interferon regulatory factor-3 by the hepatitis C virus serine protease. Science 300:1145-1148. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

