Neurite extension in central neurons: a novel role for the receptor tyrosine kinases Ror1 and Ror2
- PMID: 15654020
- PMCID: PMC1351101
- DOI: 10.1242/jcs.01622
Neurite extension in central neurons: a novel role for the receptor tyrosine kinases Ror1 and Ror2
Abstract
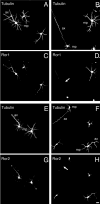
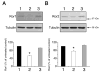
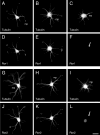
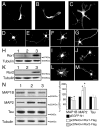
Neurite elongation and branching are key cellular events during brain development as they underlie the formation of a properly wired neuronal network. Here we report that the receptor tyrosine kinases Ror1 and Ror2 modulate the growth of neurites as well as their branching pattern in hippocampal neurons. Upon Ror1 or Ror2 suppression using antisense oligonucleotides or RNA interference (RNAi), neurons extended shorter and less branched minor processes when compared to those in control cells. In addition, Ror-depleted cells elongated longer, albeit less branched, axons than seen in control cells. Conversely, Ror overexpression both in non-neuronal cells and in hippocampal neurons resulted in the enhanced extension of short and highly branched processes. These phenotypes were accompanied by changes in the microtubule-associated proteins MAP1B and MAP2. Taken together, these results support a novel role for Ror receptors as modulators of neurite extension in central neurons.
Figures




Similar articles
-
Expression and function of the Ror-family receptor tyrosine kinases during development: lessons from genetic analyses of nematodes, mice, and humans.J Recept Signal Transduct Res. 2003 Feb;23(1):1-15. doi: 10.1081/rrs-120018757. J Recept Signal Transduct Res. 2003. PMID: 12680586 Review.
-
Ror1-Ror2 complexes modulate synapse formation in hippocampal neurons.Neuroscience. 2010 Feb 17;165(4):1261-74. doi: 10.1016/j.neuroscience.2009.11.056. Epub 2009 Dec 1. Neuroscience. 2010. PMID: 19958813 Free PMC article.
-
Differential subcellular localization of Ror tyrosine kinase receptors in cultured astrocytes.Glia. 2004 May;46(4):456-66. doi: 10.1002/glia.20023. Glia. 2004. PMID: 15095375
-
Diverse roles for the ror-family receptor tyrosine kinases in neurons and glial cells during development and repair of the nervous system.Dev Dyn. 2018 Jan;247(1):24-32. doi: 10.1002/dvdy.24515. Epub 2017 Jun 1. Dev Dyn. 2018. PMID: 28470690 Review.
-
Expression and subcellular localization of Ror tyrosine kinase receptors are developmentally regulated in cultured hippocampal neurons.J Neurosci Res. 2003 Aug 15;73(4):429-40. doi: 10.1002/jnr.10674. J Neurosci Res. 2003. PMID: 12898527
Cited by
-
Modeling Fetal Alcohol Spectrum Disorder: Validating an Ex Vivo Primary Hippocampal Cell Culture System.Alcohol Clin Exp Res. 2016 Jun;40(6):1273-82. doi: 10.1111/acer.13090. Epub 2016 May 10. Alcohol Clin Exp Res. 2016. PMID: 27162054 Free PMC article.
-
Antisera induced by infusions of autologous Ad-CD154-leukemia B cells identify ROR1 as an oncofetal antigen and receptor for Wnt5a.Proc Natl Acad Sci U S A. 2008 Feb 26;105(8):3047-52. doi: 10.1073/pnas.0712148105. Epub 2008 Feb 19. Proc Natl Acad Sci U S A. 2008. PMID: 18287027 Free PMC article.
-
Canonical and noncanonical Wnt signaling in neural stem/progenitor cells.Cell Mol Life Sci. 2015 Nov;72(21):4157-72. doi: 10.1007/s00018-015-2028-6. Epub 2015 Aug 26. Cell Mol Life Sci. 2015. PMID: 26306936 Free PMC article. Review.
-
RoR2 functions as a noncanonical Wnt receptor that regulates NMDAR-mediated synaptic transmission.Proc Natl Acad Sci U S A. 2015 Apr 14;112(15):4797-802. doi: 10.1073/pnas.1417053112. Epub 2015 Mar 30. Proc Natl Acad Sci U S A. 2015. PMID: 25825749 Free PMC article.
-
Nuclear localization of orphan receptor protein kinase (Ror1) is mediated through the juxtamembrane domain.BMC Cell Biol. 2010 Jun 30;11:48. doi: 10.1186/1471-2121-11-48. BMC Cell Biol. 2010. PMID: 20587074 Free PMC article.
References
-
- Al-Shawi R, Ashton SV, Underwood C, Simons JP. Expression of the Ror1 and Ror2 receptor tyrosine kinase genes during mouse development. Dev Genes Evol. 2001;211:161–171. - PubMed
-
- Avila J, Dominguez J, Diaz-Nido J. Regulation of microtubule dynamics by microtubule-associated protein expression and phosphorylation during neuronal development. Int J Dev Biol. 1994;38:13–25. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

