Biosynthesis of agmatine in isolated mitochondria and perfused rat liver: studies with 15N-labelled arginine
- PMID: 15656789
- PMCID: PMC1138948
- DOI: 10.1042/BJ20041260
Biosynthesis of agmatine in isolated mitochondria and perfused rat liver: studies with 15N-labelled arginine
Abstract
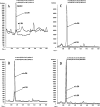
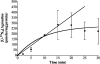
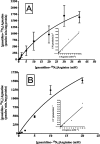
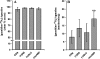
An important but unresolved question is whether mammalian mitochondria metabolize arginine to agmatine by the ADC (arginine decarboxylase) reaction. 15N-labelled arginine was used as a precursor to address this question and to determine the flux through the ADC reaction in isolated mitochondria obtained from rat liver. In addition, liver perfusion system was used to examine a possible action of insulin, glucagon or cAMP on a flux through the ADC reaction. In mitochondria and liver perfusion, 15N-labelled agmatine was generated from external 15N-labelled arginine. The production of 15N-labelled agmatine was time- and dose-dependent. The time-course of [U-15N4]agmatine formation from 2 mM [U-15N4]arginine was best fitted to a one-phase exponential curve with a production rate of approx. 29 pmol x min(-1) x (mg of protein)(-1). Experiments with an increasing concentration (0- 40 mM) of [guanidino-15N2]arginine showed a Michaelis constant Km for arginine of 46 mM and a Vmax of 3.7 nmol x min(-1) x (mg of protein)(-1) for flux through the ADC reaction. Experiments with broken mitochondria showed little changes in Vmax or Km values, suggesting that mitochondrial arginine uptake had little effect on the observed Vmax or Km values. Experiments with liver perfusion demonstrated that over 95% of the effluent agmatine was derived from perfusate [guanidino-15N2]arginine regardless of the experimental condition. However, the output of 15N-labelled agmatine (nmol x min(-1) x g(-1)) increased by approx. 2-fold (P<0.05) in perfusions with cAMP. The findings of the present study provide compelling evidence that mitochondrial ADC is present in the rat liver, and suggest that cAMP may stimulate flux through this pathway.
Figures
Similar articles
-
Regulation of urea synthesis by agmatine in the perfused liver: studies with 15N.Am J Physiol Endocrinol Metab. 2002 Dec;283(6):E1123-34. doi: 10.1152/ajpendo.00246.2002. Epub 2002 Aug 13. Am J Physiol Endocrinol Metab. 2002. PMID: 12388162
-
Putrescine biosynthesis in mammalian tissues.Biochem J. 2004 May 1;379(Pt 3):849-55. doi: 10.1042/BJ20040035. Biochem J. 2004. PMID: 14763899 Free PMC article.
-
Agmatine, a bioactive metabolite of arginine. Production, degradation, and functional effects in the kidney of the rat.J Clin Invest. 1996 Jan 15;97(2):413-20. doi: 10.1172/JCI118430. J Clin Invest. 1996. PMID: 8567962 Free PMC article.
-
Metabolic strategies for the degradation of the neuromodulator agmatine in mammals.Metabolism. 2018 Apr;81:35-44. doi: 10.1016/j.metabol.2017.11.005. Epub 2017 Nov 21. Metabolism. 2018. PMID: 29162499 Review.
-
Agmatine: multifunctional arginine metabolite and magic bullet in clinical neuroscience?Biochem J. 2017 Jul 26;474(15):2619-2640. doi: 10.1042/BCJ20170007. Biochem J. 2017. PMID: 28747403 Review.
Cited by
-
A novel bifunctional N-acetylglutamate synthase-kinase from Xanthomonas campestris that is closely related to mammalian N-acetylglutamate synthase.BMC Biochem. 2007 Apr 10;8:4. doi: 10.1186/1471-2091-8-4. BMC Biochem. 2007. PMID: 17425781 Free PMC article.
-
Expression pattern and biochemical properties of zebrafish N-acetylglutamate synthase.PLoS One. 2014 Jan 22;9(1):e85597. doi: 10.1371/journal.pone.0085597. eCollection 2014. PLoS One. 2014. PMID: 24465614 Free PMC article.
-
Structural and Mechanistic Analysis of Drosophila melanogaster Agmatine N-Acetyltransferase, an Enzyme that Catalyzes the Formation of N-Acetylagmatine.Sci Rep. 2017 Oct 18;7(1):13432. doi: 10.1038/s41598-017-13669-6. Sci Rep. 2017. PMID: 29044148 Free PMC article.
-
An arginase-based system for selection of transfected CHO cells without the use of toxic chemicals.J Biol Chem. 2019 Dec 6;294(49):18756-18768. doi: 10.1074/jbc.RA119.011162. Epub 2019 Oct 30. J Biol Chem. 2019. PMID: 31666335 Free PMC article.
-
Use of pure recombinant human enzymes to assess the disease-causing potential of missense mutations in urea cycle disorders, applied to N-acetylglutamate synthase deficiency.J Inherit Metab Dis. 2024 Nov;47(6):1194-1212. doi: 10.1002/jimd.12747. Epub 2024 May 13. J Inherit Metab Dis. 2024. PMID: 38740568 Free PMC article.
References
-
- Rasch W., Regunathan S., Li G., Reis D. J. Agmatine, the bacterial amine, is widely distributed in mammalian tissues. Life Sci. 1995;56:2319–2330. - PubMed
-
- Regunathan S., Reis D. J. Characterization of arginine decarboxylase in rat brain and liver: distinction from ornithine decarboxylase. J. Neurochem. 2000;74:2201–2208. - PubMed
-
- Feng Y., Halaris A. E., Piletz J. E. Determination of agmatine in brain and plasma using high-performance liquid chromatography with fluorescence detection. J. Chromatogr. B. 1997;691:277–286. - PubMed
-
- Gen L., Regunathan D., Barrow C. J., Esraghi J., Cooper R., Reis D. J. Agmatine: an endogenous clonidine-displacing substance in the brain. Science. 1994;263:12231–12234. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

