Desmosomal cadherin misexpression alters beta-catenin stability and epidermal differentiation
- PMID: 15657425
- PMCID: PMC544013
- DOI: 10.1128/MCB.25.3.969-978.2005
Desmosomal cadherin misexpression alters beta-catenin stability and epidermal differentiation
Abstract
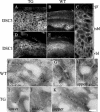
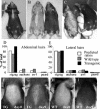
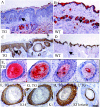
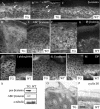
Desmosomal adhesion is important for the integrity and protective barrier function of the epidermis and is disregulated during carcinogenesis. Strong adhesion between keratinocytes is conferred by the desmosomal cadherins, desmocollin (Dsc) and desmoglein. These constitute two gene families, members of which are differentially expressed in epidermal strata. It has been suggested that this stratum-specific expression regulates keratinocyte differentiation. We tested this hypothesis by misdirecting the expression of the basally abundant desmosomal cadherins Dsc3a and Dsc3b to suprabasal differentiating keratinocytes in transgenic mice. No phenotype was apparent until adulthood, when mice developed variable ventral alopecia and had altered keratinocyte differentiation within affected areas. The follicular changes were reminiscent of changes in transgenic mice with an altered beta-catenin stability. Stabilized beta-catenin and increased beta-catenin transcriptional activity were demonstrated in transgenic mice prior to the phenotypic change and in transgenic keratinocytes as a consequence of transgene expression. Hence, a link between desmosomal cadherins and beta-catenin stability and signaling was demonstrated, and it was shown that desmocollin cadherin expression can affect keratinocyte differentiation. Furthermore, the first function for a "b-type" desmocollin cadherin was demonstrated.
Figures



References
-
- Abdalkhani, A., R. Sellers, J. Gent, H. Wulitich, S. Childress, B. Stein, R. E. Boissy, J. J. Wysolmerski, J. Foley, P. Dann, J. Hong, J. Cosgrove, B. Dreyer, D. Rimm, M. Dunbar, W. Philbrick, and J. Wysolmerski. 2002. Nipple connective tissue and its development: insights from the K14-PTHrP mouse. Mech. Dev. 115:63-77. - PubMed
-
- Chen, X., S. Bonne, M. Hatzfeld, F. van Roy, and K. J. Green. 2002. Protein binding and functional characterization of plakophilin 2. Evidence for its diverse roles in desmosomes and beta-catenin signaling. J. Biol. Chem. 277:10512-10522. - PubMed
-
- Dickson, M. A., W. C. Hahn, Y. Ino, V. Ronfard, J. Y. Wu, R. A. Weinberg, D. N. Louis, F. P. Li, and J. G. Rheinwald. 2000. Human keratinocytes that express hTERT and also bypass a p16(INK4a)-enforced mechanism that limits life span become immortal yet retain normal growth and differentiation characteristics. Mol. Cell. Biol. 20:1436-1447. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
