cAMP response element binding protein (CREB) activates transcription via two distinct genetic elements of the human glucose-6-phosphatase gene
- PMID: 15659240
- PMCID: PMC548273
- DOI: 10.1186/1471-2199-6-2
cAMP response element binding protein (CREB) activates transcription via two distinct genetic elements of the human glucose-6-phosphatase gene
Abstract
Background: The enzyme glucose-6-phosphatase catalyzes the dephosphorylation of glucose-6-phosphatase to glucose, the final step in the gluconeogenic and glycogenolytic pathways. Expression of the glucose-6-phosphatase gene is induced by glucocorticoids and elevated levels of intracellular cAMP. The effect of cAMP in regulating glucose-6-phosphatase gene transcription was corroborated by the identification of two genetic motifs CRE1 and CRE2 in the human and murine glucose-6-phosphatase gene promoter that resemble cAMP response elements (CRE).
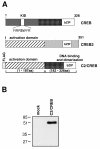
Results: The cAMP response element is a point of convergence for many extracellular and intracellular signals, including cAMP, calcium, and neurotrophins. The major CRE binding protein CREB, a member of the basic region leucine zipper (bZIP) family of transcription factors, requires phosphorylation to become a biologically active transcriptional activator. Since unphosphorylated CREB is transcriptionally silent simple overexpression studies cannot be performed to test the biological role of CRE-like sequences of the glucose-6-phosphatase gene. The use of a constitutively active CREB2/CREB fusion protein allowed us to uncouple the investigation of target genes of CREB from the variety of signaling pathways that lead to an activation of CREB. Here, we show that this constitutively active CREB2/CREB fusion protein strikingly enhanced reporter gene transcription mediated by either CRE1 or CRE2 derived from the glucose-6-phosphatase gene. Likewise, reporter gene transcription was enhanced following expression of the catalytic subunit of cAMP-dependent protein kinase (PKA) in the nucleus of transfected cells. In contrast, activating transcription factor 2 (ATF2), known to compete with CREB for binding to the canonical CRE sequence 5'-TGACGTCA-3', did not transactivate reporter genes containing CRE1, CRE2, or both CREs derived from the glucose-6-phosphatase gene.
Conclusions: Using a constitutively active CREB2/CREB fusion protein and a mutant of the PKA catalytic subunit that is targeted to the nucleus, we have shown that the glucose-6-phosphatase gene has two distinct genetic elements that function as bona fide CRE. This study further shows that the expression vectors encoding C2/CREB and catalytic subunit of PKA are valuable tools for the study of CREB-mediated gene transcription and the biological functions of CREB.
Figures



Similar articles
-
Role of basic region leucine zipper transcription factors cyclic AMP response element binding protein (CREB), CREB2, activating transcription factor 2 and CAAT/enhancer binding protein alpha in cyclic AMP response element-mediated transcription.J Neurochem. 2005 Jan;92(2):321-36. doi: 10.1111/j.1471-4159.2004.02882.x. J Neurochem. 2005. PMID: 15663480
-
Regulation of GTP cyclohydrolase I gene transcription by basic region leucine zipper transcription factors.J Cell Biochem. 2005 Dec 1;96(5):1003-20. doi: 10.1002/jcb.20580. J Cell Biochem. 2005. PMID: 16149046
-
Regulation of steroidogenesis and the steroidogenic acute regulatory protein by a member of the cAMP response-element binding protein family.Mol Endocrinol. 2002 Jan;16(1):184-99. doi: 10.1210/mend.16.1.0759. Mol Endocrinol. 2002. PMID: 11773448
-
CREB: a stimulus-induced transcription factor activated by a diverse array of extracellular signals.Annu Rev Biochem. 1999;68:821-61. doi: 10.1146/annurev.biochem.68.1.821. Annu Rev Biochem. 1999. PMID: 10872467 Review.
-
Transcription factors responsive to cAMP.Annu Rev Cell Dev Biol. 1995;11:355-77. doi: 10.1146/annurev.cb.11.110195.002035. Annu Rev Cell Dev Biol. 1995. PMID: 8689562 Review.
Cited by
-
Human nuclear pregnane X receptor cross-talk with CREB to repress cAMP activation of the glucose-6-phosphatase gene.Biochem J. 2007 Nov 1;407(3):373-81. doi: 10.1042/BJ20070481. Biochem J. 2007. PMID: 17635106 Free PMC article.
-
The long non-coding RNA Gm10768 activates hepatic gluconeogenesis by sequestering microRNA-214 in mice.J Biol Chem. 2018 Mar 16;293(11):4097-4109. doi: 10.1074/jbc.M117.812818. Epub 2018 Jan 23. J Biol Chem. 2018. PMID: 29363576 Free PMC article.
-
Regulation of Hepatic Gluconeogenesis by Nuclear Receptor Coactivator 6.Mol Cells. 2022 Apr 30;45(4):180-192. doi: 10.14348/molcells.2022.2222. Mol Cells. 2022. PMID: 35258009 Free PMC article.
-
BNDF methylation in mothers and newborns is associated with maternal exposure to war trauma.Clin Epigenetics. 2017 Jun 30;9:68. doi: 10.1186/s13148-017-0367-x. eCollection 2017. Clin Epigenetics. 2017. PMID: 28680507 Free PMC article.
-
Pterostilbene reverses palmitic acid mediated insulin resistance in HepG2 cells by reducing oxidative stress and triglyceride accumulation.Free Radic Res. 2019 Jul;53(7):815-827. doi: 10.1080/10715762.2019.1635252. Epub 2019 Jul 10. Free Radic Res. 2019. PMID: 31223033 Free PMC article.
References
-
- Foster JD, Nordlie RC. The biochemistry and molecular biology of the glucose-6-phosphatase system. Exp Biol Med. 2002;227:601–608. - PubMed
-
- Lin B, Morris DW, Chou JY. Hepatocyte nuclear factor 1 alpha is an accessory factor required for activation of glucose-6-phosphatase gene transcription by glucocorticoids. DNA Cell Biol. 1998;17:967–974. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

