Glial cell line-derived neurotrophic factor mediates the desirable actions of the anti-addiction drug ibogaine against alcohol consumption
- PMID: 15659598
- PMCID: PMC1193648
- DOI: 10.1523/JNEUROSCI.3959-04.2005
Glial cell line-derived neurotrophic factor mediates the desirable actions of the anti-addiction drug ibogaine against alcohol consumption
Abstract
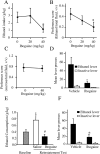
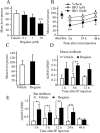
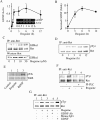
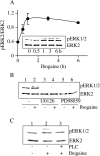
Alcohol addiction manifests as uncontrolled drinking despite negative consequences. Few medications are available to treat the disorder. Anecdotal reports suggest that ibogaine, a natural alkaloid, reverses behaviors associated with addiction including alcoholism; however, because of side effects, ibogaine is not used clinically. In this study, we first characterized the actions of ibogaine on ethanol self-administration in rodents. Ibogaine decreased ethanol intake by rats in two-bottle choice and operant self-administration paradigms. Ibogaine also reduced operant self-administration of ethanol in a relapse model. Next, we identified a molecular mechanism that mediates the desirable activities of ibogaine on ethanol intake. Microinjection of ibogaine into the ventral tegmental area (VTA), but not the substantia nigra, reduced self-administration of ethanol, and systemic administration of ibogaine increased the expression of glial cell line-derived neurotrophic factor (GDNF) in a midbrain region that includes the VTA. In dopaminergic neuron-like SHSY5Y cells, ibogaine treatment upregulated the GDNF pathway as indicated by increases in phosphorylation of the GDNF receptor, Ret, and the downstream kinase, ERK1 (extracellular signal-regulated kinase 1). Finally, the ibogaine-mediated decrease in ethanol self-administration was mimicked by intra-VTA microinjection of GDNF and was reduced by intra-VTA delivery of anti-GDNF neutralizing antibodies. Together, these results suggest that GDNF in the VTA mediates the action of ibogaine on ethanol consumption. These findings highlight the importance of GDNF as a new target for drug development for alcoholism that may mimic the effect of ibogaine against alcohol consumption but avoid the negative side effects.
Figures


References
-
- Airaksinen MS, Saarma M (2002) The GDNF family: signaling, biological functions and therapeutic value. Nat Rev Neurosci 3: 383-394. - PubMed
-
- Alper KR, Lotsof HS, Frenken GM, Luciano DJ, Bastiaans J (1999) Treatment of acute opioid withdrawal with ibogaine. Am J Addict 8: 234-242. - PubMed
-
- Bailey CP, Manley SJ, Watson WP, Wonnacott S, Molleman A, Little HJ (1998) Chronic ethanol administration alters activity in ventral tegmental area neurons after cessation of withdrawal hyperexcitability. Brain Res 803: 144-152. - PubMed
-
- Bailey CP, O'Callaghan MJ, Croft AP, Manley SJ, Little HJ (2001) Alterations in mesolimbic dopamine function during the abstinence period following chronic ethanol consumption. Neuropharmacology 41: 989-999. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
