Pyruvate:quinone oxidoreductase from Corynebacterium glutamicum: purification and biochemical characterization
- PMID: 15659664
- PMCID: PMC545707
- DOI: 10.1128/JB.187.3.862-871.2005
Pyruvate:quinone oxidoreductase from Corynebacterium glutamicum: purification and biochemical characterization
Abstract
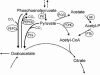
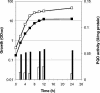
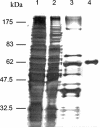
Pyruvate:quinone oxidoreductase catalyzes the oxidative decarboxylation of pyruvate to acetate and CO2 with a quinone as the physiological electron acceptor. So far, this enzyme activity has been found only in Escherichia coli. Using 2,6-dichloroindophenol as an artificial electron acceptor, we detected pyruvate:quinone oxidoreductase activity in cell extracts of the amino acid producer Corynebacterium glutamicum. The activity was highest (0.055 +/- 0.005 U/mg of protein) in cells grown on complex medium and about threefold lower when the cells were grown on medium containing glucose, pyruvate, or acetate as the carbon source. From wild-type C. glutamicum, the pyruvate:quinone oxidoreductase was purified about 180-fold to homogeneity in four steps and subjected to biochemical analysis. The enzyme is a flavoprotein, has a molecular mass of about 232 kDa, and consists of four identical subunits of about 62 kDa. It was activated by Triton X-100, phosphatidylglycerol, and dipalmitoyl-phosphatidylglycerol, and the substrates were pyruvate (kcat=37.8 +/- 3 s(-1); Km=30 +/- 3 mM) and 2-oxobutyrate (kcat=33.2 +/- 3 s(-1); Km=90 +/- 8 mM). Thiamine pyrophosphate (Km=1 microM) and certain divalent metal ions such as Mg2+ (Km=29 microM), Mn2+ (Km=2 microM), and Co2+ (Km=11 microM) served as cofactors. In addition to several dyes (2,6-dichloroindophenol, p-iodonitrotetrazolium violet, and nitroblue tetrazolium), menadione (Km=106 microM) was efficiently reduced by the purified pyruvate:quinone oxidoreductase, indicating that a naphthoquinone may be the physiological electron acceptor of this enzyme in C. glutamicum.
Figures

References
-
- Abdel-Hamid, A. M., M. M. Attwood, and J. R. Guest. 2001. Pyruvate oxidase contributes to the aerobic growth efficiency of Escherichia coli. Microbiology 147:1483-1498. - PubMed
-
- Altschul, S. F., W. Gish, W. Miller, E. W. Myers, and D. J. Lipman. 1990. Basic local alignment search tool. J. Mol. Biol. 215:403-410. - PubMed
-
- Andersson, L., H. Borg, and M. Mikaelsson. 1972. Molecular weight estimations of proteins by electrophoresis in polyacrylamide gels of graded porosity. FEBS Lett. 20:199-202. - PubMed
-
- Birnboim, H. C. 1983. A rapid alkaline extraction method for the isolation of plasmid DNA. Methods Enzymol. 100:243-255. - PubMed
-
- Blake, R., and L. P. Hager. 1978. Activation of pyruvate oxidase by monomeric and micellar amphiphiles. J. Biol. Chem. 253:1963-1971. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

