Structure of human follicle-stimulating hormone in complex with its receptor
- PMID: 15662415
- PMCID: PMC5514322
- DOI: 10.1038/nature03206
Structure of human follicle-stimulating hormone in complex with its receptor
Abstract
Follicle-stimulating hormone (FSH) is central to reproduction in mammals. It acts through a G-protein-coupled receptor on the surface of target cells to stimulate testicular and ovarian functions. We present here the 2.9-A-resolution structure of a partially deglycosylated complex of human FSH bound to the extracellular hormone-binding domain of its receptor (FSHR(HB)). The hormone is bound in a hand-clasp fashion to an elongated, curved receptor. The buried interface of the complex is large (2,600 A2) and has a high charge density. Our analysis suggests that all glycoprotein hormones bind to their receptors in this mode and that binding specificity is mediated by key interaction sites involving both the common alpha- and hormone-specific beta-subunits. On binding, FSH undergoes a concerted conformational change that affects protruding loops implicated in receptor activation. The FSH-FSHR(HB) complexes form dimers in the crystal and at high concentrations in solution. Such dimers may participate in transmembrane signal transduction.
Conflict of interest statement
Figures
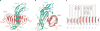

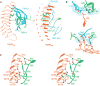
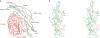
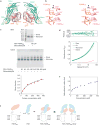
Comment in
-
Endocrinology: fertility hormone in repose.Nature. 2005 Jan 20;433(7023):203-4. doi: 10.1038/433203a. Nature. 2005. PMID: 15662395 No abstract available.
References
-
- Dias JA, et al. Molecular, structural, and cellular biology of follitropin and follitropin receptor. Vitam Horm. 2002;64:249–322. - PubMed
-
- Simoni M, Gromoll J, Nieschlag E. The follicle-stimulating hormone receptor: biochemistry, molecular biology, physiology, and pathophysiology. Endocr Rev. 1997;18:739–773. - PubMed
-
- Themmen APN, Huhtaniemi IT. Mutations of gonadotropins and gonadotropin receptors: elucidating the physiology and pathophysiology of pituitary-gonadal function. Endocr Rev. 2000;21:551–583. - PubMed
-
- Pierce JG, Parsons TF. Glycoprotein hormones: structure and function. Annu Rev Biochem. 1981;50:465–495. - PubMed
-
- Wu H, Lustbader JW, Liu Y, Canfield RE, Hendrickson WA. Structure of human chorionic gonadotropin at 2.6 Å resolution from MAD analysis of the selenomethionyl protein. Structure. 1994;2:545–558. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

