Calcium-dependent dephosphorylation of brain mitochondrial calcium/cAMP response element binding protein (CREB)
- PMID: 15663486
- PMCID: PMC2572760
- DOI: 10.1111/j.1471-4159.2004.02873.x
Calcium-dependent dephosphorylation of brain mitochondrial calcium/cAMP response element binding protein (CREB)
Abstract
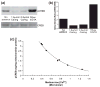
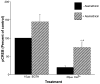
Calcium-mediated signaling regulates nuclear gene transcription by calcium/cAMP response element binding protein (CREB) via calcium-dependent kinases and phosphatases. This study tested the hypothesis that CREB is also present in mitochondria and subject to dynamic calcium-dependent modulation of its phosphorylation state. Antibodies to CREB and phosphorylated CREB (pCREB) were used to demonstrate the presence of both forms in isolated mitochondria and mitoplasts from rat brain. When energized mitochondria were exposed to increasing concentrations of Ca2+ in the physiological range, pCREB was lost while total CREB remained constant. In the presence of Ru360, an inhibitor of the mitochondrial Ca2+ uptake uniporter, calcium-dependent loss of pCREB levels was attenuated, suggesting that intramitochondrial calcium plays an important role in pCREB dephosphorylation. pCREB dephosphorylation was not, however, inhibited by the phosphatase inhibitors okadaic acid and Tacrolimus. In the absence of Ca2+, CREB phosphorylation was elevated by the addition of ATP to the mitochondrial suspension. Exposure of mitochondria to the pore-forming molecule alamethicin that causes osmotic swelling and release of intermembrane proteins enriched mitochondrial pCREB immunoreactivity. These results further suggest that mitochondrial CREB is located in the matrix or inner membrane and that a kinase and a calcium-dependent phosphatase regulate its phosphorylation state.
Figures
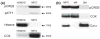
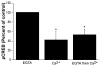

Similar articles
-
Apparent presence of Ser133-phosphorylated cyclic AMP response element binding protein (pCREB) in brain mitochondria is due to cross-reactivity of pCREB antibodies with pyruvate dehydrogenase.J Neurochem. 2005 Dec;95(5):1446-60. doi: 10.1111/j.1471-4159.2005.03471.x. Epub 2005 Oct 7. J Neurochem. 2005. PMID: 16219034
-
Activity-dependent neuroprotection and cAMP response element-binding protein (CREB): kinase coupling, stimulus intensity, and temporal regulation of CREB phosphorylation at serine 133.J Neurosci. 2005 Feb 2;25(5):1137-48. doi: 10.1523/JNEUROSCI.4288-04.2005. J Neurosci. 2005. PMID: 15689550 Free PMC article.
-
Estrogen induces phosphorylation of cyclic AMP response element binding (pCREB) in primary hippocampal cells in a time-dependent manner.Neuroscience. 2004;124(3):549-60. doi: 10.1016/j.neuroscience.2003.11.035. Neuroscience. 2004. PMID: 14980726
-
Calcium-dependent mitochondrial superoxide modulates nuclear CREB phosphorylation in hippocampal neurons.Mol Cell Neurosci. 2003 Dec;24(4):1103-15. doi: 10.1016/j.mcn.2003.09.003. Mol Cell Neurosci. 2003. PMID: 14697672
-
Control of CREB phosphorylation and its role for induction of melatonin synthesis in rat pinealocytes.Biol Cell. 1997 Nov;89(8):505-11. doi: 10.1016/s0248-4900(98)80006-3. Biol Cell. 1997. PMID: 9618900 Review.
Cited by
-
Altered Organelle Calcium Transport in Ovarian Physiology and Cancer.Cancers (Basel). 2020 Aug 10;12(8):2232. doi: 10.3390/cancers12082232. Cancers (Basel). 2020. PMID: 32785177 Free PMC article. Review.
-
The multiple functions of cytochrome c and their regulation in life and death decisions of the mammalian cell: From respiration to apoptosis.Mitochondrion. 2011 May;11(3):369-81. doi: 10.1016/j.mito.2011.01.010. Epub 2011 Feb 4. Mitochondrion. 2011. PMID: 21296189 Free PMC article. Review.
-
RNA Interference Unleashed: Current Perspective of Small Interfering RNA (siRNA) Therapeutics in the Treatment of Neuropathic Pain.ACS Pharmacol Transl Sci. 2024 Sep 23;7(10):2951-2970. doi: 10.1021/acsptsci.4c00329. eCollection 2024 Oct 11. ACS Pharmacol Transl Sci. 2024. PMID: 39416962 Review.
-
Mitochondrial mechanisms of estrogen neuroprotection.Biochim Biophys Acta. 2010 Oct;1800(10):1113-20. doi: 10.1016/j.bbagen.2009.11.013. Epub 2009 Nov 26. Biochim Biophys Acta. 2010. PMID: 19931595 Free PMC article. Review.
-
Calcium/calmodulin-dependent protein kinase IV (CaMKIV) activation contributes to the pathogenesis of experimental colitis via inhibition of intestinal epithelial cell proliferation.FASEB J. 2019 Jan;33(1):1330-1346. doi: 10.1096/fj.201800535R. Epub 2018 Aug 16. FASEB J. 2019. PMID: 30113881 Free PMC article.
References
-
- Bedogni B, Pani G, Colavitti R, Riccio A, Borello S, Murphy M, Smith R, Eboli ML, Galeotti T. Redox regulation of cAMP-responsive element-binding protein and induction of manganese superoxide dismutase in nerve growth factor-dependent cell survival. J Biol Chem. 2003;278:16 510–16 519. - PubMed
-
- Biswas G, Adebanjo OA, Freedman BD, Anandatheerthavarada HK, Vijayasarathy C, Zaidi M, Kotlikoff M, Avadhani NG. Retrograde Ca2+ signaling in C2C12 skeletal myocytes in response to mitochondrial genetic and metabolic stress: a novel mode of inter-organelle crosstalk. EMBO J. 1999;18:522–533. - PMC - PubMed
-
- Cammarota M, Paratcha G, Bevilaqua L, Levi de Stein M, Lopez M, Pellegrino de Iraldi A, Izquierdo I, Medina JH. Cyclic AMP-responsive element binding protein in brain mitochondria. J Neurochem. 1999;72:2272–2277. - PubMed
-
- Chinopoulos C, Starkov AA, Fiskum G. Cyclosporin A-insensitive permeability transition in brain mitochondria. J Biol Chem. 2003;278:27 382–27 389. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

