Differential proton sensitivity of related G protein-coupled receptors T cell death-associated gene 8 and G2A expressed in immune cells
- PMID: 15665078
- PMCID: PMC545089
- DOI: 10.1073/pnas.0409415102
Differential proton sensitivity of related G protein-coupled receptors T cell death-associated gene 8 and G2A expressed in immune cells
Abstract
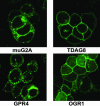
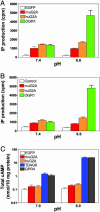
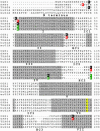
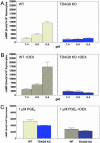
G2A, T cell death-associated gene 8 (TDAG8), ovarian cancer G protein-coupled receptor 1 (OGR1), and G protein-coupled receptor 4 (GPR4) form a group of structurally related G protein-coupled receptors (GPCRs) originally proposed to bind proinflammatory lipids. More recent studies have challenged the identification of lipid agonists for these GPCRs and have suggested that they function primarily as proton sensors. We compared the ability of these four receptors to modulate pH-dependent responses by using transiently transfected cell lines. In accordance with previously published reports, OGR1 was found to evoke strong pH-dependent responses as measured by inositol phosphate accumulation. We also confirmed the pH-dependent cAMP production by GPR4 and TDAG8. However, we found the activity of the human G2A receptor and its mouse homolog to be significantly less sensitive to pH fluctuations as measured by inositol phosphate and cAMP accumulation. Sequence homology analysis indicated that, with one exception, the histidine residues that were previously shown to be important for pH sensing by OGR1, GPR4, and TDAG8 were not conserved in the G2A receptor. We further addressed the pH-sensing properties of G2A and TDAG8 in a cellular context where these receptors are coexpressed. In thymocytes and splenocytes explanted from receptor-deficient mice, TDAG8 was found to be critical for pH-dependent cAMP production. In contrast, G2A was found to be dispensable for this process. We conclude that members of this GPCR group exhibit differential sensitivity to extracellular protons, and that expression of TDAG8 by immune cells may regulate responses in acidic microenvironments.
Figures


Similar articles
-
G2A is a proton-sensing G-protein-coupled receptor antagonized by lysophosphatidylcholine.J Biol Chem. 2004 Oct 8;279(41):42484-91. doi: 10.1074/jbc.M406561200. Epub 2004 Jul 27. J Biol Chem. 2004. PMID: 15280385
-
Involvement of proton-sensing TDAG8 in extracellular acidification-induced inhibition of proinflammatory cytokine production in peritoneal macrophages.J Immunol. 2009 Mar 1;182(5):3243-51. doi: 10.4049/jimmunol.0803466. J Immunol. 2009. PMID: 19234222
-
Heteromerization of G2A and OGR1 enhances proton sensitivity and proton-induced calcium signals.J Recept Signal Transduct Res. 2016 Dec;36(6):633-644. doi: 10.3109/10799893.2016.1155064. Epub 2016 Apr 6. J Recept Signal Transduct Res. 2016. PMID: 27049592
-
Proton-sensing and lysolipid-sensitive G-protein-coupled receptors: a novel type of multi-functional receptors.Cell Signal. 2005 Dec;17(12):1466-76. doi: 10.1016/j.cellsig.2005.06.002. Cell Signal. 2005. PMID: 16014326 Review.
-
Receptors for protons or lipid messengers or both?J Recept Signal Transduct Res. 2006;26(5-6):599-610. doi: 10.1080/10799890600932220. J Recept Signal Transduct Res. 2006. PMID: 17118800 Review.
Cited by
-
Proton-Binding Motifs of Membrane-Bound Proteins: From Bacteriorhodopsin to Spike Protein S.Front Chem. 2021 May 31;9:685761. doi: 10.3389/fchem.2021.685761. eCollection 2021. Front Chem. 2021. PMID: 34136464 Free PMC article.
-
Acidosis promotes Bcl-2 family-mediated evasion of apoptosis: involvement of acid-sensing G protein-coupled receptor Gpr65 signaling to Mek/Erk.J Biol Chem. 2012 Aug 10;287(33):27863-75. doi: 10.1074/jbc.M112.384685. Epub 2012 Jun 8. J Biol Chem. 2012. PMID: 22685289 Free PMC article.
-
Normal immune development and glucocorticoid-induced thymocyte apoptosis in mice deficient for the T-cell death-associated gene 8 receptor.Mol Cell Biol. 2006 Jan;26(2):668-77. doi: 10.1128/MCB.26.2.668-677.2006. Mol Cell Biol. 2006. PMID: 16382156 Free PMC article.
-
Lactic acidosis triggers starvation response with paradoxical induction of TXNIP through MondoA.PLoS Genet. 2010 Sep 2;6(9):e1001093. doi: 10.1371/journal.pgen.1001093. PLoS Genet. 2010. PMID: 20844768 Free PMC article.
-
GPCR activation: protonation and membrane potential.Protein Cell. 2013 Oct;4(10):747-60. doi: 10.1007/s13238-013-3073-2. Epub 2013 Sep 20. Protein Cell. 2013. PMID: 24057762 Free PMC article. Review.
References
-
- Choi, J. W., Lee, S. Y. & Choi, Y. (1996) Cell Immunol. 168, 78–84. - PubMed
-
- Xu, Y. & Casey, G. (1996) Genomics 35, 397–402. - PubMed
-
- Heiber, M., Docherty, J. M., Shah, G., Nguyen, T., Cheng, R., Heng, H. H., Marchese, A., Tsui, L. C., Shi, X., George, S. R., et al. (1995) DNA Cell Biol. 14, 25–35. - PubMed
-
- Xu, Y., Zhu, K., Hong, G., Wu, W., Baudhuin, L. M., Xiao, Y. & Damron, D. S. (2000) Nat. Cell Biol. 2, 261–267. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

