Tick saliva inhibits differentiation, maturation and function of murine bone-marrow-derived dendritic cells
- PMID: 15667568
- PMCID: PMC1782083
- DOI: 10.1111/j.1365-2567.2004.02079.x
Tick saliva inhibits differentiation, maturation and function of murine bone-marrow-derived dendritic cells
Abstract
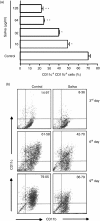
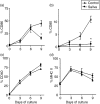
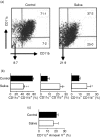
Haematophagous arthropod vectors such as mosquitoes, tsetse flies, sandflies and ticks have evolved salivary immunomodulatory factors that prevent the vertebrate host from rejecting them meanwhile enhancing pathogen transmission. As dendritic cells (DC) play a major role in host immune responses, we studied the effects of Rhipicephalus sanguineus tick saliva on DC differentiation and maturation. Flow cytometry analysis revealed that the addition of saliva to bone marrow cells inhibits the differentiation of DC and decreased the population of differentiated immature DC, increasing the levels of major histocompatibility complex (MHC) class II while not altering the expression of costimulatory (CD40, CD80 and CD86) and adhesion (CD54) molecules. Furthermore, maturation of DC stimulated by lipopolysaccharide (LPS) in the presence of saliva resulted in a lower expression of costimulatory molecules, but did not alter the up-regulation of MHC class II and CD54. The lipopolysaccharide (LPS)-matured DC cultured with saliva also presented reduced production of interleukin-12, whereas interleukin-10 production was unaltered. Assessment of the function of DC cultured with tick saliva revealed them to be poor stimulators of cytokine production by antigen-specific T cells. Our data indicate a novel modulatory role for the saliva of arthropod vectors at an initial step of the immune response through the inhibition of differentiation and maturation of DC into functional antigen-presenting cells.
Figures

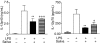
Similar articles
-
Deconstructing tick saliva: non-protein molecules with potent immunomodulatory properties.J Biol Chem. 2011 Apr 1;286(13):10960-9. doi: 10.1074/jbc.M110.205047. Epub 2011 Jan 26. J Biol Chem. 2011. PMID: 21270122 Free PMC article.
-
Yersinia enterocolitica induces apoptosis and inhibits surface molecule expression and cytokine production in murine dendritic cells.Infect Immun. 2004 Dec;72(12):7045-54. doi: 10.1128/IAI.72.12.7045-7054.2004. Infect Immun. 2004. PMID: 15557628 Free PMC article.
-
Immunosuppressive effects of Amblyomma cajennense tick saliva on murine bone marrow-derived dendritic cells.Parasit Vectors. 2015 Jan 14;8:22. doi: 10.1186/s13071-015-0634-7. Parasit Vectors. 2015. PMID: 25586117 Free PMC article.
-
Developmental aspects of dendritic cells in vitro and in vivo.Leuk Lymphoma. 1998 May;29(5-6):477-90. doi: 10.3109/10428199809050907. Leuk Lymphoma. 1998. PMID: 9643561 Review.
-
Dendritic cells in rheumatoid arthritis.Front Biosci. 2005 Jan 1;10:656-65. doi: 10.2741/1560. Print 2005 Jan 1. Front Biosci. 2005. PMID: 15569606 Review.
Cited by
-
Tick salivary compounds: their role in modulation of host defences and pathogen transmission.Front Cell Infect Microbiol. 2013 Aug 20;3:43. doi: 10.3389/fcimb.2013.00043. eCollection 2013. Front Cell Infect Microbiol. 2013. PMID: 23971008 Free PMC article. Review.
-
Salp15 binding to DC-SIGN inhibits cytokine expression by impairing both nucleosome remodeling and mRNA stabilization.PLoS Pathog. 2008 Feb 8;4(2):e31. doi: 10.1371/journal.ppat.0040031. PLoS Pathog. 2008. PMID: 18282094 Free PMC article.
-
Dendritic cells: potential triggers of autoimmunity and targets for therapy.Ann Rheum Dis. 2005 Nov;64 Suppl 4(Suppl 4):iv86-90. doi: 10.1136/ard.2005.044560. Ann Rheum Dis. 2005. PMID: 16239396 Free PMC article. Review. No abstract available.
-
Differentiated THP-1 Cells Exposed to Pathogenic and Nonpathogenic Borrelia Species Demonstrate Minimal Differences in Production of Four Inflammatory Cytokines.Vector Borne Zoonotic Dis. 2016 Nov;16(11):691-695. doi: 10.1089/vbz.2016.2006. Epub 2016 Sep 28. Vector Borne Zoonotic Dis. 2016. PMID: 27680384 Free PMC article.
-
Lead effects on development and function of bone marrow-derived dendritic cells promote Th2 immune responses.Toxicol Appl Pharmacol. 2007 Jul 1;222(1):69-79. doi: 10.1016/j.taap.2007.04.001. Epub 2007 Apr 12. Toxicol Appl Pharmacol. 2007. PMID: 17512567 Free PMC article.
References
-
- Moore J. Parasites and the behavior of biting flies. J Parasitol. 1993;79:1–16. 10.1007/BF00931210. - DOI - PubMed
-
- Jones LD, Hodgson E, Nuttall PA. Enhancement of virus transmission. By tick salivary glands. J Gen Virol. 1989;70:1895–8. - PubMed
-
- Nuttall PA. Displaced tick–parasite interactions at the host interface. Parasitology. 1998;116:S65–72. - PubMed
-
- Gillespie RD, Mbow ML, Titus RG. The immunomodulatory factors of blood-feeding arthropod saliva. Parasite Immunol. 2000;22:319–31. 10.1046/j.1365-3024.2000.00309.x. - DOI - PubMed
-
- Ribeiro JMC. Role of saliva blood-feeding by arthropods. Annu Rev Entomol. 1987;32:463–78. 10.1146/annurev.en.32.010187.002335. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

