Angular path integration by moving "hill of activity": a spiking neuron model without recurrent excitation of the head-direction system
- PMID: 15673682
- PMCID: PMC6725619
- DOI: 10.1523/JNEUROSCI.4172-04.2005
Angular path integration by moving "hill of activity": a spiking neuron model without recurrent excitation of the head-direction system
Abstract
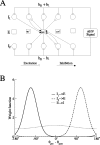
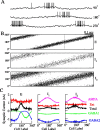
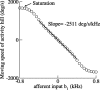
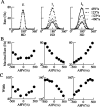
During spatial navigation, the head orientation of an animal is encoded internally by neural persistent activity in the head-direction (HD) system. In computational models, such a bell-shaped "hill of activity" is commonly assumed to be generated by recurrent excitation in a continuous attractor network. Recent experimental evidence, however, indicates that HD signal in rodents originates in a reciprocal loop between the lateral mammillary nucleus (LMN) and the dorsal tegmental nucleus (DTN), which is characterized by a paucity of local excitatory axonal collaterals. Moreover, when the animal turns its head to a new direction, the heading information is updated by a time integration of angular head velocity (AHV) signals; the underlying mechanism remains unresolved. To investigate these issues, we built and investigated an LMN-DTN network model that consists of three populations of noisy and spiking neurons coupled by biophysically realistic synapses. We found that a combination of uniform external excitation and recurrent cross-inhibition can give rise to direction-selective persistent activity. The model reproduces the experimentally observed three types of HD tuning curves differentially modulated by AHV and anticipatory firing activity in LMN HD cells. Time integration is assessed by using constant or sinusoidal angular velocity stimuli, as well as naturalistic AHV inputs (from rodent recordings). Furthermore, the internal representation of head direction is shown to be calibrated or reset by strong external cues. We identify microcircuit properties that determine the ability of our model network to subserve time integration function.
Figures






References
-
- Allen GV, Hopkins DA (1988) Mammillary body in the rat: a cytoarchitectonic, golgi, and ultrastructural study. J Comp Neurol 275: 39-64. - PubMed
-
- Allen GV, Hopkins DA (1989) Mammillary body in the rat: topography and synaptology of projections from the subicular complex, prefrontal cortex and midbrain tegmentum. J Comp Neurol 286: 311-336. - PubMed
-
- Allen GV, Hopkins DA (1990) Typography and synaptology of mamillary body projection to the mesencephalon and pons in the rat. J Comp Neurol 301: 214-231. - PubMed
-
- Bassett JP, Taube JS (2001a) Lesion of the dorsal tegmental nucleus of the rat disrupt head direction cell activity in the anterior thalamus. Soc Neurosci Abstr 27: 852.29.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
