Thalamic Cav3.1 T-type Ca2+ channel plays a crucial role in stabilizing sleep
- PMID: 15677322
- PMCID: PMC547889
- DOI: 10.1073/pnas.0409644102
Thalamic Cav3.1 T-type Ca2+ channel plays a crucial role in stabilizing sleep
Abstract
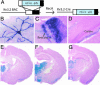
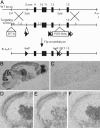
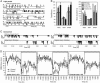
It has long been suspected that sensory signal transmission is inhibited in the mammalian brain during sleep. We hypothesized that Cav3.1 T-type Ca2+ channel currents inhibit thalamic sensory transmission to promote sleep. We found that T-type Ca2+ channel activation caused prolonged inhibition (>9 s) of action-potential firing in thalamic projection neurons of WT but not Cav3.1 knockout mice. Inhibition occurred with synaptic transmission blocked and required an increase of intracellular Ca2+. Furthermore, focal deletion of the gene encoding Cav3.1 from the rostral-midline thalamus by using Cre/loxP recombination led to frequent and prolonged arousal, which fragmented and reduced sleep. Interestingly, sleep was not disturbed when Cav3.1 was deleted from cortical pyramidal neurons. These findings support the hypothesis that thalamic T-type Ca2+ channels are required to block transmission of arousal signals through the thalamus and to stabilize sleep.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

