Plk1 docking to GRASP65 phosphorylated by Cdk1 suggests a mechanism for Golgi checkpoint signalling
- PMID: 15678101
- PMCID: PMC549625
- DOI: 10.1038/sj.emboj.7600569
Plk1 docking to GRASP65 phosphorylated by Cdk1 suggests a mechanism for Golgi checkpoint signalling
Abstract
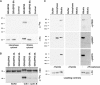
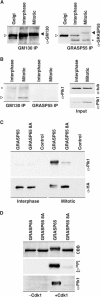
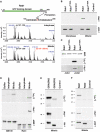
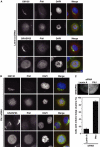
GRASP65, a structural protein of the Golgi apparatus, has been linked to the sensing of Golgi structure and the integration of this information with the control of mitotic entry in the form of a Golgi checkpoint. We show that Cdk1-cyclin B is the major kinase phosphorylating GRASP65 in mitosis, and that phosphorylated GRASP65 interacts with the polo box domain of the polo-like kinase Plk1. GRASP65 is phosphorylated in its C-terminal domain at four consensus sites by Cdk1-cyclin B, and mutation of these residues to alanine essentially abolishes both mitotic phosphorylation and Plk1 binding. Expression of the wild-type GRASP65 C-terminus but not the phosphorylation defective mutant in normal rat kidney cells causes a delay but not the block in mitotic entry expected if this were a true cell cycle checkpoint. These findings identify a Plk1-dependent signalling mechanism potentially linking Golgi structure and cell cycle control, but suggest that this may not be a cell cycle checkpoint in the classical sense.
Figures



Similar articles
-
Mitotic phosphorylation of the peripheral Golgi protein Nir2 by Cdk1 provides a docking mechanism for Plk1 and affects cytokinesis completion.Mol Cell. 2004 May 7;14(3):319-30. doi: 10.1016/s1097-2765(04)00214-x. Mol Cell. 2004. PMID: 15125835
-
Mitotic inhibition of GRASP65 organelle tethering involves Polo-like kinase 1 (PLK1) phosphorylation proximate to an internal PDZ ligand.J Biol Chem. 2010 Dec 17;285(51):39994-40003. doi: 10.1074/jbc.M110.189449. Epub 2010 Oct 11. J Biol Chem. 2010. PMID: 20937827 Free PMC article.
-
Convergence of cell cycle regulation and growth factor signals on GRASP65.J Biol Chem. 2005 Jun 17;280(24):23048-56. doi: 10.1074/jbc.M502442200. Epub 2005 Apr 15. J Biol Chem. 2005. PMID: 15834132
-
Dynamic changes in nuclear architecture during mitosis: on the role of protein phosphorylation in spindle assembly and chromosome segregation.Exp Cell Res. 1996 Dec 15;229(2):174-80. doi: 10.1006/excr.1996.0356. Exp Cell Res. 1996. PMID: 8986594 Review.
-
In search of an essential step during mitotic Golgi disassembly and inheritance.Exp Cell Res. 2001 Nov 15;271(1):22-7. doi: 10.1006/excr.2001.5367. Exp Cell Res. 2001. PMID: 11697878 Review. No abstract available.
Cited by
-
MEK1 inactivates Myt1 to regulate Golgi membrane fragmentation and mitotic entry in mammalian cells.EMBO J. 2013 Jan 9;32(1):72-85. doi: 10.1038/emboj.2012.329. Epub 2012 Dec 14. EMBO J. 2013. PMID: 23241949 Free PMC article.
-
PKD controls mitotic Golgi complex fragmentation through a Raf-MEK1 pathway.Mol Biol Cell. 2013 Feb;24(3):222-33. doi: 10.1091/mbc.E12-03-0198. Epub 2012 Dec 14. Mol Biol Cell. 2013. PMID: 23242995 Free PMC article.
-
Membrane and organelle dynamics during cell division.Nat Rev Mol Cell Biol. 2020 Mar;21(3):151-166. doi: 10.1038/s41580-019-0208-1. Epub 2020 Feb 7. Nat Rev Mol Cell Biol. 2020. PMID: 32034394 Review.
-
MLL5 maintains spindle bipolarity by preventing aberrant cytosolic aggregation of PLK1.J Cell Biol. 2016 Mar 28;212(7):829-43. doi: 10.1083/jcb.201501021. Epub 2016 Mar 21. J Cell Biol. 2016. PMID: 27002166 Free PMC article.
-
Mechanisms and Regulation of the Mitotic Inheritance of the Golgi Complex.Front Cell Dev Biol. 2015 Dec 16;3:79. doi: 10.3389/fcell.2015.00079. eCollection 2015. Front Cell Dev Biol. 2015. PMID: 26734607 Free PMC article. Review.
References
-
- Acharya U, Mallabiabarrena A, Acharya JK, Malhotra V (1998) Signaling via mitogen-activated protein kinase kinase (MEK1) is required for Golgi fragmentation during mitosis. Cell 92: 183–192 - PubMed
-
- Allan BB, Moyer BD, Balch WE (2000) Rab1 recruitment of p115 into a cis-SNARE complex: programming budding COPII vesicles for fusion. Science 289: 444–448 - PubMed
-
- Bailly E, McCaffrey M, Touchot N, Zahraoui A, Goud B, Bornens M (1991) Phosphorylation of two small GTP-binding proteins of the Rab family by p34cdc2. Nature 350: 715–718 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

