The kinase haspin is required for mitotic histone H3 Thr 3 phosphorylation and normal metaphase chromosome alignment
- PMID: 15681610
- PMCID: PMC548948
- DOI: 10.1101/gad.1267105
The kinase haspin is required for mitotic histone H3 Thr 3 phosphorylation and normal metaphase chromosome alignment
Abstract
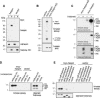
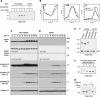
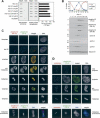
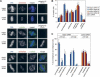
Post-translational modifications of conserved N-terminal tail residues in histones regulate many aspects of chromosome activity. Thr 3 of histone H3 is highly conserved, but the significance of its phosphorylation is unclear, and the identity of the corresponding kinase unknown. Immunostaining with phospho-specific antibodies in mammalian cells reveals mitotic phosphorylation of H3 Thr 3 in prophase and its dephosphorylation during anaphase. Furthermore we find that haspin, a member of a distinctive group of protein kinases present in diverse eukaryotes, phosphorylates H3 at Thr 3 in vitro. Importantly, depletion of haspin by RNA interference reveals that this kinase is required for H3 Thr 3 phosphorylation in mitotic cells. In addition to its chromosomal association, haspin is found at the centrosomes and spindle during mitosis. Haspin RNA interference causes misalignment of metaphase chromosomes, and overexpression delays progression through early mitosis. This work reveals a new kinase involved in composing the histone code and adds haspin to the select group of kinases that integrate regulation of chromosome and spindle function during mitosis and meiosis.
Figures



References
-
- Andrews P.D., Knatko, E., Moore, W.J., and Swedlow, J.R. 2003. Mitotic mechanics: The auroras come into view. Curr. Opin. Cell. Biol. 15: 672–683. - PubMed
-
- Barr F.A., Sillje, H.H., and Nigg, E.A. 2004. Polo-like kinases and the orchestration of cell division. Nat. Rev. Mol. Cell. Biol. 5: 429–440. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
