Inhibition of tumor necrosis factor-alpha improves physiological angiogenesis and reduces pathological neovascularization in ischemic retinopathy
- PMID: 15681845
- PMCID: PMC1602321
- DOI: 10.1016/s0002-9440(10)62284-5
Inhibition of tumor necrosis factor-alpha improves physiological angiogenesis and reduces pathological neovascularization in ischemic retinopathy
Abstract
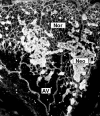
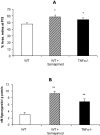
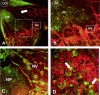
The present study was undertaken to test whether inhibition of the proangiogenic inflammatory cytokine tumor necrosis factor (TNF)-alpha can modulate retinal hypoxia and preretinal neovascularization in a murine model of oxygen-induced retinopathy (OIR). OIR was produced in TNF-alpha-/- and wild-type (WT) control C57B6 neonatal mice by exposure to 75% oxygen between postnatal days 7 and 12 (P7 to P12). Half of each WT litter was treated with the cytokine inhibitor semapimod (formerly known as CNI-1493) (5 mg/kg) by daily intraperitoneal injection from the time of reintroduction to room air at P12 until P17. The extent of preretinal neovascularization and intraretinal revascularization was quantified by image analysis of retinal flat-mounts and retinal hypoxia correlated with vascularization by immunofluorescent localization of the hypoxia-sensitive drug pimonidazole (hypoxyprobe, HP). HP adducts were also characterized by Western analysis and quantified by competitive enzyme-linked immunosorbent assay. TNF-alpha-/- and WT mice showed a similar sensitivity to hyperoxia-induced retinal ischemia at P12. At P13 some delay in early reperfusion was evident in TNF-alpha-/- and WT mice treated with semapimod. However, at P17 both these groups had significantly better vascular recovery with less ischemic/hypoxic retina and preretinal neovascularization compared to untreated retinopathy in WT mice. Immunohistochemistry showed deposition of HP in the avascular inner retina but not in areas underlying preretinal neovascularization, indicating that such aberrant vasculature can reduce retinal hypoxia. Inhibition of TNF-alpha significantly improves vascular recovery within ischemic tissue and reduces pathological neovascularization in OIR. HP provides a useful tool for mapping and quantifying tissue hypoxia in experimental ischemic retinopathy.
Figures



References
-
- McCourt M, Wang JH, Sookhai S, Redmond HP. Proinflammatory mediators stimulate neutrophil-directed angiogenesis. Arch Surg. 1999;134:1322–1331. - PubMed
-
- Leibovich SJ, Polverini PJ, Shepard HM, Wiseman DM, Shively V, Nuseir N. Macrophage-induced angiogenesis is mediated by tumour necrosis factor-alpha. Nature. 1987;329:630–632. - PubMed
-
- BenEzra D, Hemo I, Maftzir G. In vivo angiogenic activity of interleukins. Arch Ophthalmol. 1990;108:573–576. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

