Insulin-mediated down-regulation of apolipoprotein A5 gene expression through the phosphatidylinositol 3-kinase pathway: role of upstream stimulatory factor
- PMID: 15684402
- PMCID: PMC548024
- DOI: 10.1128/MCB.25.4.1537-1548.2005
Insulin-mediated down-regulation of apolipoprotein A5 gene expression through the phosphatidylinositol 3-kinase pathway: role of upstream stimulatory factor
Abstract
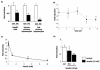
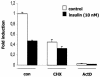
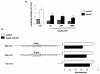
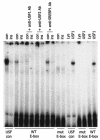
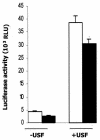
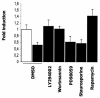
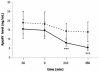
The apolipoprotein A5 gene (APOA5) has been repeatedly implicated in lowering plasma triglyceride levels. Since several studies have demonstrated that hyperinsulinemia is associated with hypertriglyceridemia, we sought to determine whether APOA5 is regulated by insulin. Here, we show that cell lines and mice treated with insulin down-regulate APOA5 expression in a dose-dependent manner. Furthermore, we found that insulin decreases human APOA5 promoter activity, and subsequent deletion and mutation analyses uncovered a functional E box in the promoter. Electrophoretic mobility shift and chromatin immunoprecipitation assays demonstrated that this APOA5 E box binds upstream stimulatory factors (USFs). Moreover, in transfection studies, USF1 stimulates APOA5 promoter activity, and the treatment with insulin reduced the binding of USF1/USF2 to the APOA5 promoter. The inhibition of the phosphatidylinositol 3-kinase (PI3K) pathway abolished insulin's effect on APOA5 gene expression, while the inhibition of the P70 S6 kinase pathway with rapamycin reversed its effect and increased APOA5 gene expression. Using an oligonucleotide precipitation assay for USF from nuclear extracts, we demonstrate that phosphorylated USF1 fails to bind to the APOA5 promoter. Taken together, these data indicate that insulin-mediated APOA5 gene transrepression could involve a phosphorylation of USFs through the PI3K and P70 S6 kinase pathways that modulate their binding to the APOA5 E box and results in APOA5 down-regulation. The effect of exogenous hyperinsulinemia in men showed a decrease in the plasma ApoAV level. These results suggest a potential contribution of the APOA5 gene in hypertriglyceridemia associated with hyperinsulinemia.
Figures


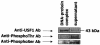
Similar articles
-
Glucose regulates the expression of the apolipoprotein A5 gene.J Mol Biol. 2008 Jul 25;380(5):789-98. doi: 10.1016/j.jmb.2008.04.057. Epub 2008 Apr 30. J Mol Biol. 2008. PMID: 18572192
-
Thyroid hormone regulates the hypotriglyceridemic gene APOA5.J Biol Chem. 2005 Jul 29;280(30):27533-43. doi: 10.1074/jbc.M503139200. Epub 2005 Jun 7. J Biol Chem. 2005. PMID: 15941710
-
Upstream stimulatory factor binding to the E-box at -65 is required for insulin regulation of the fatty acid synthase promoter.J Biol Chem. 1997 Oct 17;272(42):26367-74. doi: 10.1074/jbc.272.42.26367. J Biol Chem. 1997. PMID: 9334210
-
Is apolipoprotein A5 a novel regulator of triglyceride-rich lipoproteins?Ann Med. 2006;38(1):2-10. doi: 10.1080/07853890500407488. Ann Med. 2006. PMID: 16448983 Review.
-
Apolipoprotein A5, a newly identified gene that affects plasma triglyceride levels in humans and mice.Arterioscler Thromb Vasc Biol. 2003 Apr 1;23(4):529-34. doi: 10.1161/01.ATV.0000054194.78240.45. Epub 2002 Dec 26. Arterioscler Thromb Vasc Biol. 2003. PMID: 12615678 Review.
Cited by
-
Glucose increases hepatic lipase expression in HepG2 liver cells through upregulation of upstream stimulatory factors 1 and 2.Diabetologia. 2008 Nov;51(11):2078-87. doi: 10.1007/s00125-008-1125-6. Epub 2008 Aug 30. Diabetologia. 2008. PMID: 18758746
-
Association of the HNF1A polymorphisms and serum lipid traits, the risk of coronary artery disease and ischemic stroke.J Gene Med. 2017 Jan;19(1-2):e2941. doi: 10.1002/jgm.2941. J Gene Med. 2017. PMID: 28035729 Free PMC article.
-
Activation of hepatic lipase expression by oleic acid: possible involvement of USF1.Nutrients. 2009 Feb;1(2):133-47. doi: 10.3390/nu1020133. Epub 2009 Oct 29. Nutrients. 2009. PMID: 22253973 Free PMC article.
-
Gene-gene interaction between APOA5 and USF1: two candidate genes for the metabolic syndrome.Obes Facts. 2009;2(4):235-42. doi: 10.1159/000227288. Epub 2009 Jul 20. Obes Facts. 2009. PMID: 20054229 Free PMC article.
-
GSK3β-dependent phosphorylation alters DNA binding, transactivity and half-life of the transcription factor USF2.PLoS One. 2014 Sep 19;9(9):e107914. doi: 10.1371/journal.pone.0107914. eCollection 2014. PLoS One. 2014. PMID: 25238393 Free PMC article.
References
-
- Aouizerat, B. E., M. Kulkarni, D. Heilbron, D. Drown, S. Raskin, C. R. Pullinger, M. J. Malloy, and J. P. Kane. 2003. Genetic analysis of a polymorphism in the human apolipoprotein A-V gene: effect on plasma lipids. J. Lipid Res. 44:1167-1173. - PubMed
-
- Baroukh, N., E. Bauge, J. Akiyama, J. Chang, V. Afzal, J. C. Fruchart, E. M. Rubin, J. Fruchart, and L. A. Pennacchio. 2004. Analysis of apolipoprotein A5, C3, and plasma triglyceride concentrations in genetically engineered mice. Arterioscler. Thromb. Vasc. Biol. 24:1297-1302. - PubMed
-
- Baum, L., B. Tomlinson, and G. Thomas. 2003. APOA5-1131T>C polymorphism is associated with triglyceride levels in Chinese men. Clin. Genet. 63:377-379. - PubMed
-
- Brown, M. S., and J. L. Goldstein. 1997. The SREBP pathway: regulation of cholesterol metabolism by proteolysis of a membrane-bound transcription factor. Cell 89:331-340. - PubMed
-
- Burgering, B. M., and P. J. Coffer. 1995. Protein kinase B (c-Akt) in phosphatidylinositol-3-OH kinase signal transduction. Nature 376:599-602. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
