Activation of SOCS-3 by resistin
- PMID: 15684405
- PMCID: PMC548000
- DOI: 10.1128/MCB.25.4.1569-1575.2005
Activation of SOCS-3 by resistin
Abstract
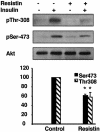
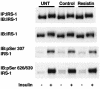
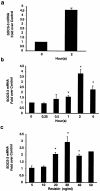
Resistin is an adipocyte hormone that modulates glucose homeostasis. Here we show that in 3T3-L1 adipocytes, resistin attenuates multiple effects of insulin, including insulin receptor (IR) phosphorylation, IR substrate 1 (IRS-1) phosphorylation, phosphatidylinositol-3-kinase (PI3K) activation, phosphatidylinositol triphosphate production, and activation of protein kinase B/Akt. Remarkably, resistin treatment markedly induces the gene expression of suppressor of cytokine signaling 3 (SOCS-3), a known inhibitor of insulin signaling. The 50% effective dose for resistin induction of SOCS-3 is approximately 20 ng/ml, close to levels of resistin in serum. Association of SOCS-3 protein with the IR is also increased by resistin. Inhibition of SOCS function prevented resistin from antagonizing insulin action in adipocytes. SOCS-3 induction is the first cellular effect of resistin that is independent of insulin and is a likely mediator of resistin's inhibitory effect on insulin signaling in adipocytes.
Figures






References
-
- Aguirre, V., E. D. Werner, J. Giraud, Y. H. Lee, S. E. Shoelson, and M. F. White. 2002. Phosphorylation of Ser307 in insulin receptor substrate-1 blocks interactions with the insulin receptor and inhibits insulin action. J. Biol. Chem. 277:1531-1537. - PubMed
-
- Azuma, K., F. Katsukawa, S. Oguchi, M. Murata, H. Yamazaki, A. Shimada, and T. Saruta. 2003. Correlation between serum resistin level and adiposity in obese individuals. Obes. Res. 11:997-1001. - PubMed
-
- Banerjee, R. R., S. M. Rangwala, J. S. Shapiro, A. S. Rich, B. Rhoades, Y. Qi, J. Wang, M. W. Rajala, A. Pocai, P. E. Scherer, C. M. Steppan, R. S. Ahima, S. Obici, L. Rossetti, and M. A. Lazar. 2004. Regulation of fasted blood glucose by resistin. Science 303:1195-1198. - PubMed
-
- Bjorbaek, C., K. El-Haschimi, J. D. Frantz, and J. S. Flier. 1999. The role of SOCS-3 in leptin signaling and leptin resistance. J. Biol. Chem. 274:30059-30065. - PubMed
-
- Bjorbaek, C., J. K. Elmquist, J. D. Frantz, S. E. Shoelson, and J. S. Flier. 1998. Identification of SOCS-3 as a potential mediator of central leptin resistance. Mol. Cell 1:619-625. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
