Injectable self-assembling peptide nanofibers create intramyocardial microenvironments for endothelial cells
- PMID: 15687132
- PMCID: PMC2754569
- DOI: 10.1161/01.CIR.0000153847.47301.80
Injectable self-assembling peptide nanofibers create intramyocardial microenvironments for endothelial cells
Abstract
Background: Promoting survival of transplanted cells or endogenous precursors is an important goal. We hypothesized that a novel approach to promote vascularization would be to create injectable microenvironments within the myocardium that recruit endothelial cells and promote their survival and organization.
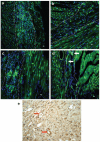
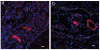
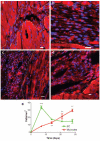
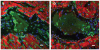
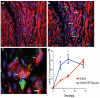
Methods and results: In this study we demonstrate that self-assembling peptides can be injected and that the resulting nanofiber microenvironments are readily detectable within the myocardium. Furthermore, the self-assembling peptide nanofiber microenvironments recruit progenitor cells that express endothelial markers, as determined by staining with isolectin and for the endothelial-specific protein platelet-endothelial cell adhesion molecule-1. Vascular smooth muscle cells are recruited to the microenvironment and appear to form functional vascular structures. After the endothelial cell population, cells that express alpha-sarcomeric actin and the transcription factor Nkx2.5 infiltrate the peptide microenvironment. When exogenous donor green fluorescent protein-positive neonatal cardiomyocytes were injected with the self-assembling peptides, transplanted cardiomyocytes in the peptide microenvironment survived and also augmented endogenous cell recruitment.
Conclusions: These experiments demonstrate that self-assembling peptides can create nanofiber microenvironments in the myocardium and that these microenvironments promote vascular cell recruitment. Because these peptide nanofibers may be modified in a variety of ways, this approach may enable injectable tissue regeneration strategies.
Figures



References
-
- Mann BK, West JL. Tissue engineering in the cardiovascular system: progress toward a tissue engineered heart. Anat Rec. 2001;263:367–371. - PubMed
-
- Li RK, Jia ZQ, Weisel RD, Mickle DA, Zhang J, Mohabeer MK, Rao V, Ivanov J. Cardiomyocyte transplantation improves heart function. Ann Thorac Surg. 1996;62:654–660. discussion 660–661. - PubMed
-
- Sakai T, Li RK, Weisel RD, Mickle DA, Kim EJ, Tomita S, Jia ZQ, Yau TM. Autologous heart cell transplantation improves cardiac function after myocardial injury. Ann Thorac Surg. 1999;68:2074–2080. discussion 2080–2081. - PubMed
-
- Papadaki M, Bursac N, Langer R, Merok J, Vunjak-Novakovic G, Freed LE. Tissue engineering of functional cardiac muscle: molecular, structural, and electrophysiological studies. Am J Physiol. 2001;280:H168–H178. - PubMed
-
- Zimmermann WH, Schneiderbanger K, Schubert P, Didie M, Munzel F, Heubach JF, Kostin S, Neuhuber WL, Eschenhagen T. Tissue engineering of a differentiated cardiac muscle construct. Circ Res. 2002;90:223–230. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

