Proteolytic cleavage of pertussis toxin S1 subunit is not essential for its activity in mammalian cells
- PMID: 15691377
- PMCID: PMC550662
- DOI: 10.1186/1471-2180-5-7
Proteolytic cleavage of pertussis toxin S1 subunit is not essential for its activity in mammalian cells
Abstract
Background: Pertussis toxin (PT) is an exotoxin virulence factor produced by Bordetella pertussis, the causative agent of whooping cough. PT consists of an active subunit (S1) that ADP-ribosylates the alpha subunit of several mammalian G proteins, and a B oligomer (S2-S5) that binds glycoconjugate receptors on cells. PT appears to enter cells by endocytosis, and retrograde transport through the Golgi apparatus may be important for its cytotoxicity. A previous study demonstrated that proteolytic processing of S1 occurs after PT enters mammalian cells. We sought to determine whether this proteolytic processing of S1 is necessary for PT cytotoxicity.
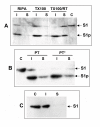
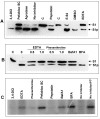
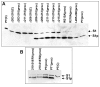
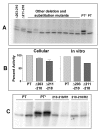
Results: Protease inhibitor studies suggested that S1 processing may involve a metalloprotease, and processing does not involve furin, a mammalian cell protease that cleaves several other bacterial toxins. However, inhibitor studies showed a general lack of correlation of S1 processing with PT cellular activity. A combination of replacement, insertion and deletion mutations in the C-terminal region of S1, as well as mass spectrometry data, suggested that the cleavage site is located around residue 203-204, but that cleavage is not strongly sequence-dependent. Processing of S1 was abolished by each of 3 overlapping 8 residue deletions just downstream of the putative cleavage site, but not by smaller deletions in the same region. Processing of the various mutant forms of PT did not correlate with cellular activity of the toxin, nor with the ability of the bacteria producing them to infect the mouse respiratory tract. In addition, S1 processing was not detected in transfected cells expressing S1, even though S1 was fully active in these cells.
Conclusions: S1 processing is not essential for the cellular activity of PT. This distinguishes it from the processing of various other bacterial toxins, which has been shown to be important for their cytotoxicity. S1 processing may be mediated primarily by a metalloprotease, but the cleavage site on S1 is not sequence-dependent and processing appears to depend on the general topology of the protein in that region, indicating that multiple proteases may contribute to this cleavage.
Figures



Similar articles
-
Expression, activity and cytotoxicity of pertussis toxin S1 subunit in transfected mammalian cells.Cell Microbiol. 2001 Jan;3(1):45-54. doi: 10.1046/j.1462-5822.2001.00092.x. Cell Microbiol. 2001. PMID: 11207619
-
Evading the proteasome: absence of lysine residues contributes to pertussis toxin activity by evasion of proteasome degradation.Infect Immun. 2007 Jun;75(6):2946-53. doi: 10.1128/IAI.02011-06. Epub 2007 Apr 9. Infect Immun. 2007. PMID: 17420233 Free PMC article.
-
Monoclonal antibody that neutralizes pertussis toxin activities.Asian Pac J Allergy Immunol. 2007 Mar;25(1):37-45. Asian Pac J Allergy Immunol. 2007. PMID: 17891920
-
The ins and outs of pertussis toxin.FEBS J. 2011 Dec;278(23):4668-82. doi: 10.1111/j.1742-4658.2011.08237.x. Epub 2011 Aug 4. FEBS J. 2011. PMID: 21740523 Review.
-
A proposed mechanism of ADP-ribosylation catalyzed by the pertussis toxin S1 subunit.Biochimie. 1995;77(5):333-40. doi: 10.1016/0300-9084(96)88143-0. Biochimie. 1995. PMID: 8527486 Review.
Cited by
-
Intracellular disassembly and activity of pertussis toxin require interaction with ATP.Pathog Dis. 2016 Aug;74(6):ftw065. doi: 10.1093/femspd/ftw065. Epub 2016 Jun 30. Pathog Dis. 2016. PMID: 27369899 Free PMC article.
-
Bordetella pertussis infection exacerbates influenza virus infection through pertussis toxin-mediated suppression of innate immunity.PLoS One. 2011 Apr 20;6(4):e19016. doi: 10.1371/journal.pone.0019016. PLoS One. 2011. PMID: 21533103 Free PMC article.
-
Cloning and Expression of S1 Subunit of Pertussis Toxin in Escherichia coli.Avicenna J Med Biotechnol. 2011 Jan;3(1):19-24. Avicenna J Med Biotechnol. 2011. PMID: 23407437 Free PMC article.
-
Evidence of accelerated evolution and ectodermal-specific expression of presumptive BDS toxin cDNAs from Anemonia viridis.Mar Drugs. 2013 Oct 30;11(11):4213-31. doi: 10.3390/md11114213. Mar Drugs. 2013. PMID: 24177670 Free PMC article.
-
The pertussis toxin S1 subunit is a thermally unstable protein susceptible to degradation by the 20S proteasome.Biochemistry. 2006 Nov 21;45(46):13734-40. doi: 10.1021/bi061175+. Biochemistry. 2006. PMID: 17105192 Free PMC article.
References
-
- Tamura M, Nogimori L, Murai S, Yajima M, Itio K, Katada T, Ui M, Ishii S. Subunit structure of the islet-activating protein, pertussis toxin, in conformity with the A-B model. Biochemistry. 1982;21:5516–5522. - PubMed
-
- Moss J, Stanley SJ, Burns DL, Hsia JA, Yost DA, Myers GA, Hewlett EL. Activation by thiol of the latent NAD glycohydrolase and ADP-ribosyltransferase activities of Bordetella pertussis toxin (islet-activating protein) J Biol Chem. 1983;258:11879–11882. - PubMed
-
- Brennan MJ, David JL, Kenimer JG, Manclark CR. Lectin-like binding of pertussis toxin to a 165 kilodalton Chinese hamster ovary cell glycoprotein. J Biol Chem. 1988;263:4895–4899. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

