Characterization of a novel human sperm-associated antigen 9 (SPAG9) having structural homology with c-Jun N-terminal kinase-interacting protein
- PMID: 15693750
- PMCID: PMC1184539
- DOI: 10.1042/BJ20041577
Characterization of a novel human sperm-associated antigen 9 (SPAG9) having structural homology with c-Jun N-terminal kinase-interacting protein
Abstract
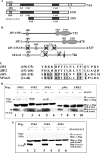
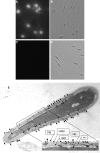
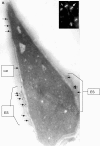
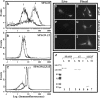
We report a novel SPAG9 (sperm-associated antigen 9) protein having structural homology with JNK (c-Jun N-terminal kinase)-interacting protein 3. SPAG9, a single copy gene mapped to the human chromosome 17q21.33 syntenic with location of mouse chromosome 11, was earlier shown to be expressed exclusively in testis [Shankar, Mohapatra and Suri (1998) Biochem. Biophys. Res. Commun. 243, 561-565]. The SPAG9 amino acid sequence analysis revealed identity with the JNK-binding domain and predicted coiled-coil, leucine zipper and transmembrane domains. The secondary structure analysis predicted an alpha-helical structure for SPAG9 that was confirmed by CD spectra. Microsequencing of higher-order aggregates of recombinant SPAG9 by tandem MS confirmed the amino acid sequence and mono atomic mass of 83.9 kDa. Transient expression of SPAG9 and its deletion mutants revealed that both leucine zipper with extended coiled-coil domains and transmembrane domain of SPAG9 were essential for dimerization and proper localization. Studies of MAPK (mitogenactivated protein kinase) interactions demonstrated that SPAG9 interacted with higher binding affinity to JNK3 and JNK2 compared with JNK1. No interaction was observed with p38alpha or extracellular-signal-regulated kinase pathways. Polyclonal antibodies raised against recombinant SPAG9 recognized native protein in human sperm extracts and localized specifically on the acrosomal compartment of intact human spermatozoa. Acrosome-reacted spermatozoa demonstrated SPAG9 immunofluorescence, indicating its retention on the equatorial segment after the acrosome reaction. Further, anti-SPAG9 antibodies inhibited the binding of human spermatozoa to intact human oocytes as well as to matched hemizona. This is the first report of sperm-associated JNK-binding protein that may have a role in spermatozoa-egg interaction.
Figures



Similar articles
-
Isolation and characterization of a haploid germ cell specific sperm associated antigen 9 (SPAG9) from the baboon.Mol Reprod Dev. 2004 Oct;69(2):186-93. doi: 10.1002/mrd.20164. Mol Reprod Dev. 2004. PMID: 15293220
-
Molecular cloning and characterization of the macaque sperm associated antigen 9 (SPAG9): an orthologue of human SPAG9 gene.Mol Reprod Dev. 2005 May;71(1):58-66. doi: 10.1002/mrd.20245. Mol Reprod Dev. 2005. PMID: 15736136
-
Sperm associated antigen 9 (SPAG9): a new member of c-Jun NH2 -terminal kinase (JNK) interacting protein exclusively expressed in testis.Keio J Med. 2005 Jun;54(2):66-71. doi: 10.2302/kjm.54.66. Keio J Med. 2005. PMID: 16077255
-
Immunogenicity and contraceptive potential of recombinant human sperm associated antigen (SPAG9).J Reprod Immunol. 2005 Oct;67(1-2):69-76. doi: 10.1016/j.jri.2005.06.004. Epub 2005 Sep 6. J Reprod Immunol. 2005. PMID: 16143403 Review.
-
Emerging role of sperm-associated antigen 9 in tumorigenesis.Biomed Pharmacother. 2018 Jul;103:1212-1216. doi: 10.1016/j.biopha.2018.04.168. Epub 2018 May 7. Biomed Pharmacother. 2018. PMID: 29864900 Review.
Cited by
-
Cancer testis antigens: A new paradigm for cancer therapy.Oncoimmunology. 2012 Oct 1;1(7):1194-1196. doi: 10.4161/onci.20686. Oncoimmunology. 2012. PMID: 23170277 Free PMC article.
-
Overexpression of SPAG9 correlates with poor prognosis and tumor progression in hepatocellular carcinoma.Tumour Biol. 2014 Aug;35(8):7685-91. doi: 10.1007/s13277-014-2030-x. Epub 2014 May 8. Tumour Biol. 2014. PMID: 24801907
-
SPAG9 is involved in hepatocarcinoma cell migration and invasion via modulation of ELK1 expression.Onco Targets Ther. 2016 Mar 1;9:1067-75. doi: 10.2147/OTT.S98727. eCollection 2016. Onco Targets Ther. 2016. PMID: 27042099 Free PMC article.
-
Sperm associated antigen 9 promotes oncogenic KSHV-encoded interferon regulatory factor-induced cellular transformation and angiogenesis by activating the JNK/VEGFA pathway.PLoS Pathog. 2020 Aug 10;16(8):e1008730. doi: 10.1371/journal.ppat.1008730. eCollection 2020 Aug. PLoS Pathog. 2020. PMID: 32776977 Free PMC article.
-
SPAG9 is overexpressed in osteosarcoma, and regulates cell proliferation and invasion through regulation of JunD.Oncol Lett. 2016 Oct;12(4):2674-2679. doi: 10.3892/ol.2016.4920. Epub 2016 Jul 29. Oncol Lett. 2016. PMID: 27698841 Free PMC article.
References
-
- Evans J. P., Florman H. M. The state of the union: the cell biology of fertilization. Nat. Cell Biol. 2002;4(Suppl.):S57–S63. - PubMed
-
- Singson A., Zannoni S., Kadandale P. Molecules that function in the steps of fertilization. Cytokine Growth Factor Rev. 2001;12:299–304. - PubMed
-
- Vacquier V. D. Evolution of gamete recognition proteins. Science. 1998;281:1995–1998. - PubMed
-
- Yanagimachi R. Mammalian fertilization. In: Knobil E., Neill J. D., editors. The Physiology of Reproduction. New York: Raven Press; 1994. pp. 189–317.
-
- Talbot P., Shur B. D., Myles D. G. Cell adhesion and fertilization: steps in oocyte transport, sperm-zona pellucida interactions, and sperm-egg fusion. Biol. Reprod. 2003;68:1–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

