Stability and specificity of heterodimer formation for the coiled-coil neck regions of the motor proteins Kif3A and Kif3B: the role of unstructured oppositely charged regions
- PMID: 15705165
- PMCID: PMC1403826
- DOI: 10.1111/j.1399-3011.2005.00210.x
Stability and specificity of heterodimer formation for the coiled-coil neck regions of the motor proteins Kif3A and Kif3B: the role of unstructured oppositely charged regions
Abstract
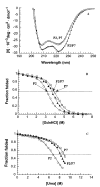
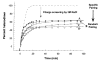
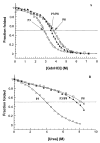
We investigated the folding, stability, and specificity of dimerization of the neck regions of the kinesin-like proteins Kif3A (residues 356-416) and Kif3B (residues 351-411). We showed that the complementary charged regions found in the hinge regions (which directly follow the neck regions) of these proteins do not adopt any secondary structure in solution. We then explored the ability of the complementary charged regions to specify heterodimer formation for the neck region coiled-coils found in Kif3A and Kif3B. Redox experiments demonstrated that oppositely charged regions specified the formation of a heterodimeric coiled-coil. Denaturation studies with urea demonstrated that the negatively charged region of Kif3A dramatically destabilized its neck coiled-coil (urea1/2 value of 3.9 m compared with 6.7 m for the coiled-coil alone). By comparison, the placement of a positively charged region C-terminal to the neck coiled-coil of Kif3B had little effect on stability (urea1/2 value of 8.2 m compared with 8.8 m for the coiled-coil alone). The pairing of complementary charged regions leads to specific heterodimer formation where the stability of the heterodimeric neck coiled-coil with charged regions had similar stability (urea1/2 value of 7.8 m) to the most stable homodimer (Kif3B) with charged regions (urea1/2 value of 8.0 m) and dramatically more stable than the Kif3A homodimer with charged regions (urea1/2, value of 3.9 m). The heterodimeric coiled-coil with charged extensions has essentially the same stability as the heterodimeric coiled-coil on its own (urea1/2 values of 7.8 and 8.1 m, respectively) suggesting that specificity of heterodimerization is driven by non-specific attraction of the oppositely unstructured charged regions without affecting stability of the heterodimeric coiled-coil.
Figures





Similar articles
-
The role of unstructured highly charged regions on the stability and specificity of dimerization of two-stranded alpha-helical coiled-coils: analysis of the neck-hinge region of the kinesin-like motor protein Kif3A.J Struct Biol. 2002 Jan-Feb;137(1-2):206-19. doi: 10.1006/jsbi.2002.4446. J Struct Biol. 2002. PMID: 12064947
-
Helix capping interactions stabilize the N-terminus of the kinesin neck coiled-coil.J Struct Biol. 2002 Jan-Feb;137(1-2):220-35. doi: 10.1006/jsbi.2002.4475. J Struct Biol. 2002. PMID: 12064948
-
Identification of kinesin neck region as a stable alpha-helical coiled coil and its thermodynamic characterization.Biochemistry. 1997 Feb 18;36(7):1933-42. doi: 10.1021/bi962392l. Biochemistry. 1997. PMID: 9048581
-
Use of a heterodimeric coiled-coil system for biosensor application and affinity purification.J Chromatogr B Biomed Sci Appl. 1998 Sep 11;715(1):307-29. doi: 10.1016/s0378-4347(98)00172-8. J Chromatogr B Biomed Sci Appl. 1998. PMID: 9792518 Review.
-
Coiled-coils: stability, specificity, and drug delivery potential.Adv Drug Deliv Rev. 2002 Oct 18;54(8):1113-29. doi: 10.1016/s0169-409x(02)00058-3. Adv Drug Deliv Rev. 2002. PMID: 12384310 Review.
Cited by
-
How kinesin-2 forms a stalk.Mol Biol Cell. 2011 Nov;22(22):4279-87. doi: 10.1091/mbc.E11-02-0112. Epub 2011 Sep 14. Mol Biol Cell. 2011. PMID: 21917588 Free PMC article.
-
A mutual co-recognition mechanism ensures the proper assembly of heterotrimeric kinesin-2 for intraflagellar transport.Nat Commun. 2025 Jul 24;16(1):6816. doi: 10.1038/s41467-025-62152-8. Nat Commun. 2025. PMID: 40707480 Free PMC article.
-
Intrinsic Disorder in the Kinesin Superfamily.Biophys Rev. 2013 Sep 1;5(3):233-47. doi: 10.1007/s12551-012-0096-5. Biophys Rev. 2013. PMID: 24244223 Free PMC article.
-
Kinesin-2 motors: Kinetics and biophysics.J Biol Chem. 2018 Mar 23;293(12):4510-4518. doi: 10.1074/jbc.R117.001324. Epub 2018 Feb 14. J Biol Chem. 2018. PMID: 29444824 Free PMC article. Review.
-
The translocation selectivity of the kinesins that mediate neuronal organelle transport.Traffic. 2012 Apr;13(4):549-64. doi: 10.1111/j.1600-0854.2011.01325.x. Epub 2012 Jan 24. Traffic. 2012. PMID: 22212743 Free PMC article.
References
-
- Bloom GS, Wagner MC, Pfister KK, Brady ST. Native structure and physical properties of bovine brain kinesin and identification of the ATP-binding subunit polypeptide. Biochemistry. 1988;27:3409–3416. - PubMed
-
- Hirokawa N, Pfister KK, Yorifuji H, Wagner MC, Brady ST, Bloom GS. Submolecular domains of bovine brain kinesin identified by electron microscopy and monoclonal antibody decoration. Cell. 1989;56:867–878. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

