Engineered G protein coupled receptors reveal independent regulation of internalization, desensitization and acute signaling
- PMID: 15707483
- PMCID: PMC551613
- DOI: 10.1186/1741-7007-3-3
Engineered G protein coupled receptors reveal independent regulation of internalization, desensitization and acute signaling
Abstract
Background: The physiological regulation of G protein-coupled receptors, through desensitization and internalization, modulates the length of the receptor signal and may influence the development of tolerance and dependence in response to chronic drug treatment. To explore the importance of receptor regulation, we engineered a series of Gi-coupled receptors that differ in signal length, degree of agonist-induced internalization, and ability to induce adenylyl cyclase superactivation. All of these receptors, based on the kappa opioid receptor, were modified to be receptors activated solely by synthetic ligands (RASSLs). This modification allows us to compare receptors that have the same ligands and effectors, but differ only in desensitization and internalization.
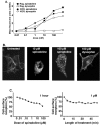
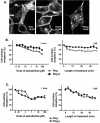
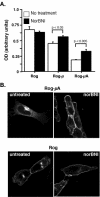
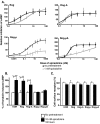
Results: Removal of phosphorylation sites in the C-terminus of the RASSL resulted in a mutant that was resistant to internalization and less prone to desensitization. Replacement of the C-terminus of the RASSL with the corresponding portion of the mu opioid receptor eliminated the induction of AC superactivation, without disrupting agonist-induced desensitization or internalization. Surprisingly, removal of phosphorylation sites from this chimera resulted in a receptor that is constitutively internalized, even in the absence of agonist. However, the receptor still signals and desensitizes in response to agonist, indicating normal G-protein coupling and partial membrane expression.
Conclusions: These studies reveal that internalization, desensitization and adenylyl cyclase superactivation, all processes that decrease chronic Gi-receptor signals, are independently regulated. Furthermore, specific mutations can radically alter superactivation or internalization without affecting the efficacy of acute Gi signaling. These mutant RASSLs will be useful for further elucidating the temporal dynamics of the signaling of G protein-coupled receptors in vitro and in vivo.
Figures

Similar articles
-
Relationship between the G protein signaling and homologous desensitization of G protein-coupled receptors.Biochem Biophys Res Commun. 2006 Jan 13;339(2):695-700. doi: 10.1016/j.bbrc.2005.11.070. Biochem Biophys Res Commun. 2006. PMID: 16325780
-
Conditional expression and signaling of a specifically designed Gi-coupled receptor in transgenic mice.Nat Biotechnol. 1999 Feb;17(2):165-9. doi: 10.1038/6165. Nat Biotechnol. 1999. PMID: 10052353
-
Agonist-induced regulation and trafficking of kappa opioid receptors.Life Sci. 2004 Jun 18;75(5):511-36. doi: 10.1016/j.lfs.2003.10.041. Life Sci. 2004. PMID: 15158363 Review.
-
Ultra-low-dose naloxone suppresses opioid tolerance, dependence and associated changes in mu opioid receptor-G protein coupling and Gbetagamma signaling.Neuroscience. 2005;135(1):247-61. doi: 10.1016/j.neuroscience.2005.06.003. Neuroscience. 2005. PMID: 16084657
-
The role of mu opioid receptor desensitization and endocytosis in morphine tolerance and dependence.Curr Opin Neurobiol. 2007 Oct;17(5):556-64. doi: 10.1016/j.conb.2007.10.004. Epub 2007 Dec 18. Curr Opin Neurobiol. 2007. PMID: 18068348 Review.
Cited by
-
Constitutive Gs activation using a single-construct tetracycline-inducible expression system in embryonic stem cells and mice.Stem Cell Res Ther. 2011 Mar 4;2(2):11. doi: 10.1186/scrt52. Stem Cell Res Ther. 2011. PMID: 21375737 Free PMC article.
-
Engineering GPCR signaling pathways with RASSLs.Nat Methods. 2008 Aug;5(8):673-8. doi: 10.1038/nmeth.1232. Nat Methods. 2008. PMID: 18668035 Free PMC article. Review.
-
Gs G protein-coupled receptor signaling in osteoblasts elicits age-dependent effects on bone formation.J Bone Miner Res. 2010 Mar;25(3):584-93. doi: 10.1002/jbmr.3. J Bone Miner Res. 2010. PMID: 20200944 Free PMC article.
-
Osteoblast expression of an engineered Gs-coupled receptor dramatically increases bone mass.Proc Natl Acad Sci U S A. 2008 Jan 29;105(4):1209-14. doi: 10.1073/pnas.0707457105. Epub 2008 Jan 22. Proc Natl Acad Sci U S A. 2008. PMID: 18212126 Free PMC article.
-
The Anti-Cancer Effect of A3 Adenosine Receptor Agonists: A Novel, Targeted Therapy.Immunol Endocr Metab Agents Med Chem. 2007 Aug;7:298-303. doi: 10.2174/187152207781369878. Immunol Endocr Metab Agents Med Chem. 2007. PMID: 34824647 Free PMC article.
References
-
- Ferguson SSG. Evolving concepts in G protein-coupled receptor endocytosis: The role in receptor desensitization and signaling. Pharmacol Rev. 2001;53:1–24. - PubMed

