A CCAAT/enhancer binding protein beta isoform, liver-enriched inhibitory protein, regulates commitment of osteoblasts and adipocytes
- PMID: 15713650
- PMCID: PMC549359
- DOI: 10.1128/MCB.25.5.1971-1979.2005
A CCAAT/enhancer binding protein beta isoform, liver-enriched inhibitory protein, regulates commitment of osteoblasts and adipocytes
Abstract
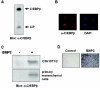
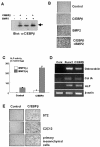
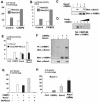
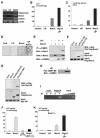
Although both osteoblasts and adipocytes have a common origin, i.e., mesenchymal cells, the molecular mechanisms that define the direction of two different lineages are presently unknown. In this study, we investigated the role of a transcription factor, CCAAT/enhancer binding protein beta (C/EBPbeta), and its isoform in the regulation of balance between osteoblast and adipocyte differentiation. We found that C/EBPbeta, which is induced along with osteoblast differentiation, promotes the differentiation of mesenchymal cells into an osteoblast lineage in cooperation with Runx2, an essential transcription factor for osteogenesis. Surprisingly, an isoform of C/EBPbeta, liver-enriched inhibitory protein (LIP), which lacks the transcriptional activation domain, stimulates transcriptional activity and the osteogenic action of Runx2, although LIP inhibits adipogenesis in a dominant-negative fashion. Furthermore, LIP physically associates with Runx2 and binds to the C/EBP binding element present in the osteocalcin gene promoter. These data indicate that LIP functions as a coactivator for Runx2 and preferentially promotes the osteoblast differentiation of mesenchymal cells. Thus, identification of a novel role of the C/EBPbeta isoform provides insight into the molecular basis of the regulation of osteoblast and adipocyte commitment.
Figures

References
-
- Ahrens, M., T. Ankenbauer, D. Schroder, A. Hollnagel, H. Mayer, and G. Gross. 1993. Expression of human bone morphogenetic proteins-2 or -4 in murine mesenchymal progenitor C3H10T1/2 cells induces differentiation into distinct mesenchymal cell lineages. DNA Cell Biol. 12:871-880. - PubMed
-
- Akune, T., S. Ohba, S. Kamekura, M. Yamaguchi, U. I. Chung, N. Kubota, Y. Terauchi, Y. Harada, Y. Azuma, K. Nakamura, T. Kadowaki, and H. Kawaguchi. 2004. PPARgamma insufficiency enhances osteogenesis through osteoblast formation from bone marrow progenitors. J. Clin. Investig. 113:846-855. - PMC - PubMed
-
- Bachner, D., M. Ahrens, D. Schroder, A. Hoffmann, J. Lauber, N. Betat, P. Steinert, L. Flohe, and G. Gross. 1998. Bmp-2 downstream targets in mesenchymal development identified by subtractive cloning from recombinant mesenchymal progenitors (C3H10T1/2). Dev. Dyn. 213:398-411. - PubMed
-
- Blake, T., N. Adya, C. H. Kim, A. C. Oates, L. Zon, A. Chitnis, B. M. Weinstein, and P. P. Liu. 2000. Zebrafish homolog of the leukemia gene CBFB: its expression during embryogenesis and its relationship to scl and gata-1 in hematopoiesis. Blood 96:4178-4184. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
