Monoclonal antibody 16D10 to the C-terminal domain of the feto-acinar pancreatic protein binds to membrane of human pancreatic tumoral SOJ-6 cells and inhibits the growth of tumor xenografts
- PMID: 15720797
- PMCID: PMC1531675
- DOI: 10.1593/neo.04298
Monoclonal antibody 16D10 to the C-terminal domain of the feto-acinar pancreatic protein binds to membrane of human pancreatic tumoral SOJ-6 cells and inhibits the growth of tumor xenografts
Abstract
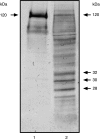
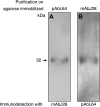
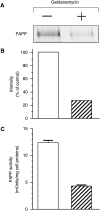
Feto-acinar pancreatic protein (FAPP) characterized by mAbJ28 reactivity is a specific component associated with ontogenesis and behaves as an oncodevelopment-associated antigen. We attempted to determine whether pancreatic tumoral SOJ-6 cells are expressed at their surface FAPP antigens and to examine if specific antibodies directed against these FAPP epitopes could decrease the growth of pancreatic tumors in a mice model. For this purpose, we used specific antibodies against either the whole FAPP, the O-glycosylated C-terminal domain, or the N-terminal domain of the protein. Our results indicate that SOJ-6 cells expressed at their surface a 32-kDa peptide corresponding to the C-terminal domain of the FAPP. Furthermore, we show, by using endoproteinase Lys-C or geldanamycin, a drug able to impair the FAPP secretion, that this 32-kDa peptide expressed on the SOJ-6 cell surface comes from the degradation of the FAPP. Finally, an in vivo prospective study using a preventative tumor model in nude mice indicates that targeting this peptide by the use of mAb16D10 inhibits the growth of SOJ-6 xenografts. The specificity of mAb16D10 for pancreatic tumors and the possibility to obtain recombinant structures of mucin-like peptides recognized by mAb16D10 and mAbJ28 are promising tools in immunologic approaches to cure pancreatic cancers.
Figures


Similar articles
-
The mucinous domain of pancreatic carboxyl-ester lipase (CEL) contains core 1/core 2 O-glycans that can be modified by ABO blood group determinants.J Biol Chem. 2018 Dec 14;293(50):19476-19491. doi: 10.1074/jbc.RA118.001934. Epub 2018 Oct 12. J Biol Chem. 2018. PMID: 30315106 Free PMC article.
-
Monoclonal antibody 16D10 to the COOH-terminal domain of the feto-acinar pancreatic protein targets pancreatic neoplastic tissues.Mol Cancer Ther. 2009 Feb;8(2):282-91. doi: 10.1158/1535-7163.MCT-08-0471. Epub 2009 Feb 3. Mol Cancer Ther. 2009. PMID: 19190122
-
Targeting a novel onco-glycoprotein antigen at tumoral pancreatic cell surface by mAb16D10 induces cell death.J Immunol. 2012 Oct 1;189(7):3386-96. doi: 10.4049/jimmunol.1102647. Epub 2012 Sep 5. J Immunol. 2012. PMID: 22956586
-
The formation of the oncofetal J28 glycotope involves core-2 beta6-N-acetylglucosaminyltransferase and alpha3/4-fucosyltransferase activities.Glycobiology. 1999 Sep;9(9):935-46. doi: 10.1093/glycob/9.9.935. Glycobiology. 1999. PMID: 10460835
-
Glycoengineering of alphaGal xenoantigen on recombinant peptide bearing the J28 pancreatic oncofetal glycotope.Glycobiology. 2007 Jun;17(6):620-30. doi: 10.1093/glycob/cwm028. Epub 2007 Mar 20. Glycobiology. 2007. PMID: 17374617
Cited by
-
Blind SELEX Approach Identifies RNA Aptamers That Regulate EMT and Inhibit Metastasis.Mol Cancer Res. 2017 Jul;15(7):811-820. doi: 10.1158/1541-7786.MCR-16-0462. Epub 2017 Apr 10. Mol Cancer Res. 2017. PMID: 28396463 Free PMC article.
-
Therapeutic antibodies for the treatment of pancreatic cancer.ScientificWorldJournal. 2010 Jun 15;10:1107-20. doi: 10.1100/tsw.2010.103. ScientificWorldJournal. 2010. PMID: 20563534 Free PMC article. Review.
-
The mucinous domain of pancreatic carboxyl-ester lipase (CEL) contains core 1/core 2 O-glycans that can be modified by ABO blood group determinants.J Biol Chem. 2018 Dec 14;293(50):19476-19491. doi: 10.1074/jbc.RA118.001934. Epub 2018 Oct 12. J Biol Chem. 2018. PMID: 30315106 Free PMC article.
-
Investigation of a new tumor-associated glycosylated antigen as target for dendritic cell vaccination in pancreatic cancer.Oncoimmunology. 2012 Jan 1;1(1):56-61. doi: 10.4161/onci.1.1.18459. Oncoimmunology. 2012. PMID: 22720212 Free PMC article.
-
Exosomal lipids impact notch signaling and induce death of human pancreatic tumoral SOJ-6 cells.PLoS One. 2012;7(10):e47480. doi: 10.1371/journal.pone.0047480. Epub 2012 Oct 19. PLoS One. 2012. PMID: 23094054 Free PMC article.
References
-
- Lombardo D. Bile salt-dependent lipase: its pathophysiological implications. Biochim Biophys Acta. 2001;1533:1–28. - PubMed
-
- Lombardo D, Guy O. Studies on the substrate specificity of a carboxyl ester hydrolase from human pancreatic juice: II. Action on cholesterol esters and lipid-soluble vitamin esters. Biochim Biophys Acta. 1980;611:147–155. - PubMed
-
- Shamir R, Johnson WJ, Zolfaghari R, Lee HS, Fisher EA. Role of bile salt-dependent cholesteryl ester hydrolase in the uptake of micellar cholesterol by intestinal cells. Biochemistry. 1995;34:6351–6358. - PubMed
-
- Howles P, Carter C, Hui DY. Dietary free and esterified cholesterol absorption in cholesterol esterase (bile salt-stimulated lipase) gene-targeted mice. J Biol Chem. 1996;271:7196–7202. - PubMed
-
- Bruneau N, Lombardo D. Chaperone function of a Grp 94-related protein for folding and transport of the pancreatic bile salt-dependent lipase. J Biol Chem. 1995;270:13524–13533. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
