Recombinant guinea pig tumor necrosis factor alpha stimulates the expression of interleukin-12 and the inhibition of Mycobacterium tuberculosis growth in macrophages
- PMID: 15731034
- PMCID: PMC1064954
- DOI: 10.1128/IAI.73.3.1367-1376.2005
Recombinant guinea pig tumor necrosis factor alpha stimulates the expression of interleukin-12 and the inhibition of Mycobacterium tuberculosis growth in macrophages
Abstract
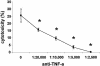
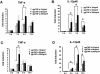
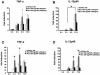
Tumor necrosis factor alpha (TNF-alpha) plays an important role in the host immune response to infection with the intracellular pathogen Mycobacterium tuberculosis. It is essential for the formation of protective tuberculous granulomas and regulates the expression of other cytokines which contribute to a protective immune response. Interleukin-12 (IL-12) is known to promote a Th1 response, which is essential for antimycobacterial resistance. Recombinant guinea pig TNF-alpha (rgpTNF-alpha) protein (17 kDa) was purified, and its bioactivity was confirmed by its cytotoxicity for L929 fibroblasts. High titers of polyclonal anti-gpTNF-alpha antibody were obtained by immunization of rabbits. Resident alveolar and peritoneal macrophages were isolated from guinea pigs and infected with either the H37Ra or H37Rv strain of M. tuberculosis. The mRNA levels for TNF-alpha and IL-12 p40 were measured using real-time PCR. IL-12 p40 mRNA was up-regulated in a dose-dependent manner by rgpTNF-alpha alone. In infected macrophages, a lower dose of rgpTNF-alpha intensified the mRNA levels of TNF-alpha and IL-12 p40. However, higher doses of rgpTNF-alpha suppressed TNF-alpha and IL-12 p40 mRNA. The antimycobacterial activity of macrophages was assessed by metabolic labeling of M. tuberculosis with [3H]uracil. Resident alveolar and peritoneal macrophages treated with anti-gpTNF-alpha antibody to block endogenous TNF-alpha exhibited increased intracellular mycobacterial growth. These data suggest that the dose of TNF-alpha is crucial to the stimulation of optimal expression of protective cytokines and that TNF-alpha contributes to the control of mycobacterial replication to promote host resistance against M. tuberculosis.
Figures



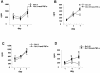
References
-
- Barrera, L. F., E. Skamene, and D. Radzioch. 1993. Assessment of mycobacterial infection and multiplication in macrophages by polymerase chain reaction. J. Immunol. Methods 157:91-99. - PubMed
-
- Bekker, L. G., G. Maartens, L. Steyn, and, G., Kaplan. 1998. Selective increase in plasma tumor necrosis factor-alpha and concomitant clinical deterioration after initiating therapy in patients with severe tuberculosis. J. Infect. Dis. 178:580-584. - PubMed
-
- Boehm, U., T. Klamp, M. Groot, and J. C. Howard. 1997. Cellular responses to interferon-gamma. Annu. Rev. Immunol. 15:749-795. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

