Protein-DNA charge transport: redox activation of a DNA repair protein by guanine radical
- PMID: 15738421
- PMCID: PMC553321
- DOI: 10.1073/pnas.0409410102
Protein-DNA charge transport: redox activation of a DNA repair protein by guanine radical
Abstract
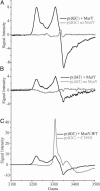
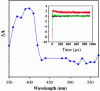
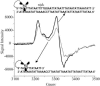
DNA charge transport (CT) chemistry provides a route to carry out oxidative DNA damage from a distance in a reaction that is sensitive to DNA mismatches and lesions. Here, DNA-mediated CT also leads to oxidation of a DNA-bound base excision repair enzyme, MutY. DNA-bound Ru(III), generated through a flash/quench technique, is found to promote oxidation of the [4Fe-4S](2+) cluster of MutY to [4Fe-4S](3+) and its decomposition product [3Fe-4S](1+). Flash/quench experiments monitored by EPR spectroscopy reveal spectra with g = 2.08, 2.06, and 2.02, characteristic of the oxidized clusters. Transient absorption spectra of poly(dGC) and [Ru(phen)(2)dppz](3+) (dppz = dipyridophenazine), generated in situ, show an absorption characteristic of the guanine radical that is depleted in the presence of MutY with formation instead of a long-lived species with an absorption at 405 nm; we attribute this absorption also to formation of the oxidized [4Fe-4S](3+) and [3Fe-4S](1+) clusters. In ruthenium-tethered DNA assemblies, oxidative damage to the 5'-G of a 5'-GG-3' doublet is generated from a distance but this irreversible damage is inhibited by MutY and instead EPR experiments reveal cluster oxidation. With ruthenium-tethered assemblies containing duplex versus single-stranded regions, MutY oxidation is found to be mediated by the DNA duplex, with guanine radical as an intermediate oxidant; guanine radical formation facilitates MutY oxidation. A model is proposed for the redox activation of DNA repair proteins through DNA CT, with guanine radicals, the first product under oxidative stress, in oxidizing the DNA-bound repair proteins, providing the signal to stimulate DNA repair.
Figures


References
-
- Hall, D. B., Holmlin, R. E. & Barton, J. K. (1996) Nature 382, 731-735. - PubMed
-
- Hall, D. B., Kelley, S. O. & Barton, J. K. (1998) Biochemistry 37, 15933-15940. - PubMed
-
- Gasper, S. M. & Schuster, G. B. (1997) J. Am. Chem. Soc. 119, 12762-12771.
-
- Meggers, E., Kusch, D., Spichty, M., Wille, U. & Giese, B. (1998) Angew. Chem. Int. Ed. 37, 460-462. - PubMed
-
- Nakatani, K., Dohno, C. & Saito, I. (1999) J. Am. Chem. Soc. 121, 10854-10855.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

