The role of regulatory T cells in antigen-induced arthritis: aggravation of arthritis after depletion and amelioration after transfer of CD4+CD25+ T cells
- PMID: 15743476
- PMCID: PMC1065322
- DOI: 10.1186/ar1484
The role of regulatory T cells in antigen-induced arthritis: aggravation of arthritis after depletion and amelioration after transfer of CD4+CD25+ T cells
Abstract
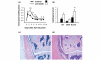
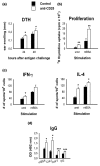
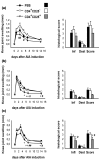
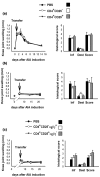
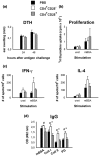
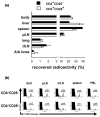
It is now generally accepted that CD4+CD25+ Treg cells play a major role in the prevention of autoimmunity and pathological immune responses. Their involvement in the pathogenesis of chronic arthritis is controversial, however, and so we examined their role in experimental antigen-induced arthritis in mice. Depletion of CD25-expressing cells in immunized animals before arthritis induction led to increased cellular and humoral immune responses to the inducing antigen (methylated bovine serum albumin; mBSA) and autoantigens, and to an exacerbation of arthritis, as indicated by clinical (knee joint swelling) and histological scores. Transfer of CD4+CD25+ cells into immunized mice at the time of induction of antigen-induced arthritis decreased the severity of disease but was not able to cure established arthritis. No significant changes in mBSA-specific immune responses were detected. In vivo migration studies showed a preferential accumulation of CD4+CD25+ cells in the inflamed joint as compared with CD4+CD25- cells. These data imply a significant role for CD4+CD25+ Treg cells in the control of chronic arthritis. However, transferred Treg cells appear to be unable to counteract established acute or chronic inflammation. This is of considerable importance for the timing of Treg cell transfer in potential therapeutic applications.
Figures

References
-
- Kinne RW, Palombo-Kinne E, Emmrich F. T-cells in the pathogenesis of rheumatoid arthritis villains or accomplices? Biochim Biophys Acta. 1997;1360:109–141. - PubMed
-
- Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol. 1995;155:1151–1164. - PubMed
-
- Stephens LA, Mason D. CD25 is a marker for CD4+ thymocytes that prevent autoimmune diabetes in rats, but peripheral T cells with this function are found in both CD25+ and CD25- subpopulations. J Immunol. 2000;165:3105–3110. - PubMed
-
- Baecher-Allan C, Brown JA, Freeman GJ, Hafler DA. CD4+CD25high regulatory cells in human peripheral blood. J Immunol. 2001;167:1245–1253. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

