Elevated endothelial nitric oxide bioactivity and resistance to angiotensin-dependent hypertension in 12/15-lipoxygenase knockout mice
- PMID: 15743778
- PMCID: PMC1602346
- DOI: 10.1016/S0002-9440(10)62287-0
Elevated endothelial nitric oxide bioactivity and resistance to angiotensin-dependent hypertension in 12/15-lipoxygenase knockout mice
Abstract
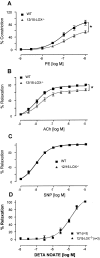
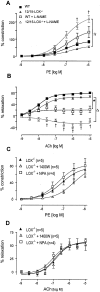
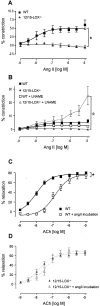
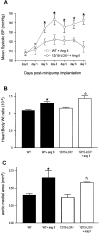
12/15-Lipoxygenase (12/15-LOX) plays a pathogenic role in atherosclerosis. To characterize whether 12/15-LOX also contributes to endothelial dysfunction and hypertension, regulation of vessel tone and angiotensin II (ang II) responses were characterized in mice deficient in 12/15-LOX. There was a twofold increase in the magnitude of l-nitroarginine-methyl ester-inhibitable, acetylcholine-dependent relaxation or phenylephrine-dependent constriction in aortic rings isolated from 12/15-LOX(-/-) mice. Plasma NO metabolites and aortic endothelial NO synthase (eNOS) expression were also elevated twofold. Angiotensin II failed to vasoconstrict 12/15-LOX(-/-) aortic rings in the absence of L-nitroarginine-methyl ester, and ang II impaired acetylcholine-induced relaxation in wild-type, but not 12/15-LOX(-/-) rings. In vivo, 12/15-LOX(-/-) mice had similar basal systolic blood pressure measurements to wild type, however, blood pressure elevations in response to ang II infusion (1.1 mg/kg/day) were significantly attenuated (maximal pressure, 143.4 +/- 4 mmHg versus 122.1 +/- 5.3 mmHg for wild type and 12/15-LOX(-/-), respectively). In contrast, vascular hypertrophic responses to ang II, and ang II type 1 receptor (AT1-R) expression were similar in both strains. This study shows that 12/15-LOX(-/-) mice have increased NO biosynthesis and impaired ang II-dependent vascular responses in vitro and in vivo, suggesting that 12/15-LOX signaling contributes to impaired NO bioactivity in vascular disease in vivo.
Figures



References
-
- Kuhn H, Thiele BJ. The diversity of the lipoxygenase family. Many sequence data but little information on biological significance. FEBS Lett. 1999;449:7–11. - PubMed
-
- Brash AR. Lipoxygenases: occurrence, functions, catalysis, and acquisition of substrate. J Biol Chem. 1999;274:23679–23682. - PubMed
-
- Yla-Herttuala S, Rosenfeld ME, Parthasarathy S, Sigal E, Sarkioja T, Witztum JL, Steinberg D. Gene expression in macrophage-rich human atherosclerotic lesions. 15-Lipoxygenase and acetyl low density lipoprotein receptor messenger RNA colocalize with oxidation specific lipid-protein adducts. J Clin Invest. 1991;87:1146–1152. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

