Cross-talk between Schwann cells and neuroblasts influences the biology of neuroblastoma xenografts
- PMID: 15743800
- PMCID: PMC1602341
- DOI: 10.1016/S0002-9440(10)62309-7
Cross-talk between Schwann cells and neuroblasts influences the biology of neuroblastoma xenografts
Abstract
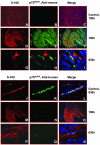
Neuroblastoma (NB) tumors with abundant schwannian stroma have a differentiated phenotype, low vascularity, and are associated with a favorable prognosis. These observations suggest that cross-talk between Schwann cells and neuroblasts may influence tumor biology. To test this hypothesis, we developed a novel NB xenograft model with infiltrating mouse Schwann cells. Human SMS-KCNR NB cells were injected intrafascicularly (sciatic nerve-engrafted NB, n = 19) or outside the sciatic nerve (control, n = 12). Xenografts were harvested 4 to 12 weeks after tumor cell inoculation for histological studies. Schwann cells were immunostained with S-100 and species-specific p75(NGFR), major histocompatibility complex, and human leukocyte antigen antibodies. The number of proliferating cells, infiltrating Schwann cells, apoptotic cells, differentiated neuroblasts, and blood vessels in the sciatic nerve-engrafted NB tumors were compared to controls. Significantly more Schwann cells were detected in the sciatic nerve-engrafted NB xenografts than controls (P < 0.001). The infiltrating Schwann cells were S-100-positive and reacted with anti-mouse major histocompatibility complex class Ib and p75(NGFR) but not anti-human p75(NGFR) and human leukocyte antigen class I antibodies. The sciatic nerve-engrafted tumors also had lower numbers of proliferating neuroblasts, higher numbers of differentiated neuroblasts and apoptotic cells, and decreased vascular density compared to controls. Our results indicate that infiltrating Schwann cells of mouse origin are capable of promoting human neuroblast differentiation, inducing apoptosis, and inhibiting proliferation and angiogenesis in vivo.
Figures




Similar articles
-
'Cross-talk' between Schwannian stroma and neuroblasts promotes neuroblastoma tumor differentiation and inhibits angiogenesis.Cancer Lett. 2005 Oct 18;228(1-2):125-31. doi: 10.1016/j.canlet.2005.01.056. Cancer Lett. 2005. PMID: 15935552 Review.
-
Presence of cancer-associated fibroblasts inversely correlates with Schwannian stroma in neuroblastoma tumors.Mod Pathol. 2009 Jul;22(7):950-8. doi: 10.1038/modpathol.2009.52. Epub 2009 May 1. Mod Pathol. 2009. PMID: 19407854 Free PMC article.
-
Schwann cell-conditioned medium inhibits angiogenesis.Cancer Res. 2000 Nov 1;60(21):5966-71. Cancer Res. 2000. PMID: 11085514
-
Schwannian cell differentiation of human neuroblastoma cell lines in vitro induced by bromodeoxyuridine.Cancer Res. 1988 May 1;48(9):2531-7. Cancer Res. 1988. PMID: 2833349
-
Schwann cells in neuroblastoma.Eur J Cancer. 1995;31A(4):429-34. doi: 10.1016/0959-8049(95)00051-j. Eur J Cancer. 1995. PMID: 7576940 Review.
Cited by
-
Cell-type-specific transcriptomics in chimeric models using transcriptome-based masks.Nucleic Acids Res. 2005 Jul 19;33(13):e111. doi: 10.1093/nar/gni104. Nucleic Acids Res. 2005. PMID: 16030348 Free PMC article.
-
Microencapsulation of Neuroblastoma Cells and Mesenchymal Stromal Cells in Collagen Microspheres: A 3D Model for Cancer Cell Niche Study.PLoS One. 2015 Dec 14;10(12):e0144139. doi: 10.1371/journal.pone.0144139. eCollection 2015. PLoS One. 2015. PMID: 26657086 Free PMC article.
-
Neuropeptide Y as a Biomarker and Therapeutic Target for Neuroblastoma.Am J Pathol. 2016 Nov;186(11):3040-3053. doi: 10.1016/j.ajpath.2016.07.019. Epub 2016 Oct 12. Am J Pathol. 2016. PMID: 27743558 Free PMC article.
-
Truncated DNMT3B isoform DNMT3B7 suppresses growth, induces differentiation, and alters DNA methylation in human neuroblastoma.Cancer Res. 2012 Sep 15;72(18):4714-23. doi: 10.1158/0008-5472.CAN-12-0886. Epub 2012 Jul 18. Cancer Res. 2012. PMID: 22815530 Free PMC article.
-
Wnt Signalling Drives Context-Dependent Differentiation or Proliferation in Neuroblastoma.Neoplasia. 2018 Apr;20(4):335-350. doi: 10.1016/j.neo.2018.01.009. Epub 2018 Mar 3. Neoplasia. 2018. PMID: 29505958 Free PMC article.
References
-
- Brodeur GM, Maris JM. Neuroblastoma. Pizzo PA, Poplack DG, editors. Philadelphia: Lippincott-Raven,; Principles and Practice of Pediatric Oncology. 2001:pp 895–937.
-
- Brodeur GM. Neuroblastoma: biological insights into a clinical enigma. Nat Rev Cancer. 2003;3:203–216. - PubMed
-
- Maris JM, Matthay KK. Molecular biology of neuroblastoma. J Clin Oncol. 1999;17:2226–2279. - PubMed
-
- Shimada H, Chatten J, Newton WA, Jr, Sachs N, Hamoudi AB, Chiba T, Marsden HB, Misugi K. Histopathologic prognostic factors in neuroblastic tumors: definition of subtypes of ganglioneuroblastoma and an age-linked classification of neuroblastomas. J Natl Cancer Inst. 1984;73:405–416. - PubMed
-
- Shimada H, Ambros IM, Dehner LP, Hata J, Joshi VV, Roald B, Stram DO, Gerbing RB, Lukens JN, Matthay KK, Castleberry RP. The International Neuroblastoma Pathology Classification (the Shimada System). Cancer. 1999;86:364–372. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

