Genomic imprinting of XX spermatogonia and XX oocytes recovered from XX<-->XY chimeric testes
- PMID: 15746241
- PMCID: PMC554793
- DOI: 10.1073/pnas.0406769102
Genomic imprinting of XX spermatogonia and XX oocytes recovered from XX<-->XY chimeric testes
Abstract
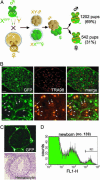
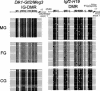
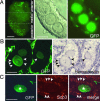
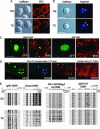
We produced XX<-->XY chimeras by using embryos whose X chromosomes were tagged with EGFP (X*), making the fluorescent green female (XX*) germ cells easily distinguishable from their nonfluorescent male (XY) counterparts. Taking advantage of tagging with EGFP, the XX* "prospermatogonia" were isolated from the testes, and the status of their genomic imprinting was examined. It was shown that these XX cells underwent a paternal imprinting, despite their chromosomal constitution. As previously indicated in sex-reversal XXsxr testes, we also found a few green XX* germ cells developed as "eggs" within the seminiferous tubules of XX*<-->XY chimeric testes. These cells were indistinguishable from XX* prospermatogonia at birth but resumed oogenesis in a testicular environment. The biological nature of the "testicular eggs" was examined by recovering the eggs from chimeric testes. The testicular eggs not only formed an egg-specific structure, the zona pellucida, but also were able to fuse with sperm. The collected testicular eggs were indicated to undergo maternal imprinting, despite the testicular environment. The genomic imprinting did not always follow the environmental conditions of where the germ cells resided; rather, it was defined by the sex that was chosen by the germ cells at early embryonic stage.
Figures
Similar articles
-
Sex reversal of genetic females (XX) induced by the transplantation of XY somatic cells in the medaka, Oryzias latipes.Int J Dev Biol. 2002 Aug;46(5):711-7. Int J Dev Biol. 2002. PMID: 12216983
-
Potency of testicular somatic environment to support spermatogenesis in XX/Sry transgenic male mice.Development. 2007 Feb;134(3):449-54. doi: 10.1242/dev.02751. Epub 2006 Dec 21. Development. 2007. PMID: 17185318
-
Live-borns from XX but not XY oocytes in the chimeric mouse ovary composed of B6.Y(TIR) and XX cells.Biol Reprod. 1998 Feb;58(2):574-82. doi: 10.1095/biolreprod58.2.574. Biol Reprod. 1998. PMID: 9475416
-
The role of sex chromosomes in mammalian germ cell differentiation: can the germ cells carrying X and Y chromosomes differentiate into fertile oocytes?Asian J Androl. 2015 May-Jun;17(3):360-6. doi: 10.4103/1008-682X.143306. Asian J Androl. 2015. PMID: 25578929 Free PMC article. Review.
-
Sex determination and sex reversal: genotype, phenotype, dogma and semantics.Hum Genet. 1992 Jul;89(5):467-79. doi: 10.1007/BF00219168. Hum Genet. 1992. PMID: 1634224 Review.
Cited by
-
Mammalian haploid stem cells: establishment, engineering and applications.Cell Mol Life Sci. 2019 Jun;76(12):2349-2367. doi: 10.1007/s00018-019-03069-6. Epub 2019 Mar 19. Cell Mol Life Sci. 2019. PMID: 30888429 Free PMC article. Review.
-
Aberrant gene expression and sexually incompatible genomic imprinting in oocytes derived from XY mouse embryonic stem cells in vitro.PLoS One. 2013;8(3):e58555. doi: 10.1371/journal.pone.0058555. Epub 2013 Mar 5. PLoS One. 2013. PMID: 23472205 Free PMC article.
-
Analysis of the sperm flagellar axoneme using gene-modified mice.Exp Anim. 2020 Nov 12;69(4):374-381. doi: 10.1538/expanim.20-0064. Epub 2020 Jun 18. Exp Anim. 2020. PMID: 32554934 Free PMC article. Review.
-
Molecular biological features of male germ cell differentiation.Reprod Med Biol. 2007 Feb 16;6(1):1-9. doi: 10.1111/j.1447-0578.2007.00158.x. eCollection 2007 Mar. Reprod Med Biol. 2007. PMID: 29699260 Free PMC article. Review.
-
Haploid embryonic stem cell lines derived from androgenetic and parthenogenetic rat blastocysts.J Reprod Dev. 2017 Dec 15;63(6):611-616. doi: 10.1262/jrd.2017-074. Epub 2017 Aug 19. J Reprod Dev. 2017. PMID: 28824040 Free PMC article.
References
-
- Merchant-Larios, H. & Moreno-Mendoza, N. (2001) Arch. Med. Res. 32, 553–558. - PubMed
-
- Lee, C. H. & Taketo, T. (2001) Genesis 30, 7–11. - PubMed
-
- Lovell-Badge, R. & Robertson, E. (1990) Development (Cambridge, U.K.) 109, 635–646. - PubMed
-
- Koopman, P., Gubbay, J., Vivian, N., Goodfellow, P. & Lovell-Badge, R. (1991) Nature 351, 117–121. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

