G-protein signaling: back to the future
- PMID: 15747061
- PMCID: PMC2794341
- DOI: 10.1007/s00018-004-4462-3
G-protein signaling: back to the future
Abstract
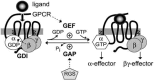
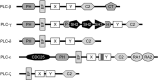
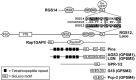
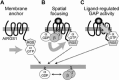
Heterotrimeric G-proteins are intracellular partners of G-protein-coupled receptors (GPCRs). GPCRs act on inactive Galpha.GDP/Gbetagamma heterotrimers to promote GDP release and GTP binding, resulting in liberation of Galpha from Gbetagamma. Galpha.GTP and Gbetagamma target effectors including adenylyl cyclases, phospholipases and ion channels. Signaling is terminated by intrinsic GTPase activity of Galpha and heterotrimer reformation - a cycle accelerated by 'regulators of G-protein signaling' (RGS proteins). Recent studies have identified several unconventional G-protein signaling pathways that diverge from this standard model. Whereas phospholipase C (PLC) beta is activated by Galpha(q) and Gbetagamma, novel PLC isoforms are regulated by both heterotrimeric and Ras-superfamily G-proteins. An Arabidopsis protein has been discovered containing both GPCR and RGS domains within the same protein. Most surprisingly, a receptor-independent Galpha nucleotide cycle that regulates cell division has been delineated in both Caenorhabditis elegans and Drosophila melanogaster. Here, we revisit classical heterotrimeric G-protein signaling and explore these new, non-canonical G-protein signaling pathways.
Figures




References
-
- Evanko D. S., Thiyagarajan M. M., Siderovski D. P. and Wedegaertner P. B. (2001) Gbeta gamma isoforms selectively rescue plasma membrane localization and palmitoylation of mutant Galphas and Galphaq. J. Biol. Chem. 276: 23945–23953 - PubMed
-
- Chen C. A. and Manning D. R. (2001) Regulation of G proteins by covalent modification. Oncogene 20: 1643–1652 - PubMed
-
- Robillard L., Ethier N., Lachance M. and Hebert T. E. (2000) Gbetagamma subunit combinations differentially modulate receptor and effector coupling in vivo. Cell Signal. 12: 673–682 - PubMed
-
- Brandt D. R. and Ross E. M. (1985) GTPase activity of the stimulatory GTP-binding regulatory protein of adenylate cyclase, Gs. Accumulation and turnover of enzyme-nucleotide intermediates. J. Biol. Chem. 260: 266–272 - PubMed
-
- Higashijima T., Ferguson K. M., Sternweis P. C., Smigel M. D. and Gilman A. G. (1987) Effects of Mg2+ and the beta gamma-subunit complex on the interactions of guanine nucleotides with G proteins. J. Biol. Chem. 262: 762–766 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

