Epithelial tight junction proteins as potential antibody targets for pancarcinoma therapy
- PMID: 15750830
- PMCID: PMC11036788
- DOI: 10.1007/s00262-004-0613-x
Epithelial tight junction proteins as potential antibody targets for pancarcinoma therapy
Abstract
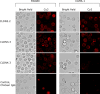
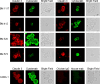
Recombinant monoclonal antibodies are beginning to revolutionize cancer therapy. In combination with standard chemotherapy, high response rates have been reported with antibodies of the human IgG1 isotype for treatment of non-Hodgkin's lymphoma and breast cancer. It is becoming apparent that targets for antibody-based therapies do not necessarily need to be absent from normal tissues but can be present there either in low copy numbers or with binding epitopes shielded from the therapeutic antibody. Here, we studied whether claudin proteins that form tight junctions in normal epithelia are still expressed on carcinoma cells and whether their extracellular domains can be recognized by antibodies. We show that mRNAs of claudins 1, 3, 4, and 7 are all expressed in different human carcinoma cell lines, while claudin 8 was selectively expressed in breast and pancreas cancer lines. Chicken polyclonal antibodies were raised against peptides contained within predicted extracellular domains of claudins 1, 3, and 4. Affinity-purified IgG fractions for claudins 3 and 4 were monospecific and bound to human breast and colon carcinoma lines, but not to a line of monocytic origin. Claudin 3 antibodies also homogeneously stained human renal cell carcinoma tissue and micrometastatic tumor cells as identified by cytokeratin staining in bone marrow biopsies of breast cancer patients. Fluorescence-activated cell sorting and immunocytochemistry indicated that claudin antibodies bound to the surface of tumor cells. By analogy to other tumor-associated antigens that are differentially accessible to antibodies on tumor vs normal tissue, we propose that certain claudin proteins have potential as targets for novel antibody-based therapies of carcinomas.
Figures




Similar articles
-
Heterogeneity in expression and subcellular localization of claudins 2, 3, 4, and 5 in the rat liver, pancreas, and gut.Gastroenterology. 2001 Feb;120(2):411-22. doi: 10.1053/gast.2001.21736. Gastroenterology. 2001. PMID: 11159882
-
Claudin extracellular domains determine paracellular charge selectivity and resistance but not tight junction fibril architecture.Am J Physiol Cell Physiol. 2003 Jun;284(6):C1346-54. doi: 10.1152/ajpcell.00547.2002. Epub 2003 Apr 16. Am J Physiol Cell Physiol. 2003. PMID: 12700140
-
Endothelial claudin: claudin-5/TMVCF constitutes tight junction strands in endothelial cells.J Cell Biol. 1999 Oct 4;147(1):185-94. doi: 10.1083/jcb.147.1.185. J Cell Biol. 1999. PMID: 10508865 Free PMC article.
-
Ruffles and spikes: Control of tight junction morphology and permeability by claudins.Biochim Biophys Acta Biomembr. 2020 Sep 1;1862(9):183339. doi: 10.1016/j.bbamem.2020.183339. Epub 2020 May 7. Biochim Biophys Acta Biomembr. 2020. PMID: 32389670 Free PMC article. Review.
-
The tumor antigen EpCAM: tetraspanins and the tight junction protein claudin-7, new partners, new functions.Front Biosci. 2008 May 1;13:5847-65. doi: 10.2741/3121. Front Biosci. 2008. PMID: 18508627 Review.
Cited by
-
Estrogen-responsive genes overlap with triiodothyronine-responsive genes in a breast carcinoma cell line.ScientificWorldJournal. 2014 Jan 23;2014:969404. doi: 10.1155/2014/969404. eCollection 2014. ScientificWorldJournal. 2014. PMID: 24587767 Free PMC article.
-
Role of Estrogen Receptor-α in the Regulation of Claudin-6 Expression in Breast Cancer Cells.J Breast Cancer. 2011 Mar;14(1):20-7. doi: 10.4048/jbc.2011.14.1.20. Epub 2011 Mar 31. J Breast Cancer. 2011. PMID: 21847390 Free PMC article.
-
Claudins in Cancer: A Current and Future Therapeutic Target.Int J Mol Sci. 2024 Apr 24;25(9):4634. doi: 10.3390/ijms25094634. Int J Mol Sci. 2024. PMID: 38731853 Free PMC article. Review.
-
A novel screening system for claudin binder using baculoviral display.PLoS One. 2011 Feb 14;6(2):e16611. doi: 10.1371/journal.pone.0016611. PLoS One. 2011. PMID: 21339813 Free PMC article.
-
Tight junctions: a barrier to the initiation and progression of breast cancer?J Biomed Biotechnol. 2010;2010:460607. doi: 10.1155/2010/460607. Epub 2009 Nov 15. J Biomed Biotechnol. 2010. PMID: 19920867 Free PMC article. Review.
References
-
- Apostolopoulos V, McKenzie IF. Cellular mucins: targets for immunotherapy. Crit Rev Immunol. 1994;14:293–309. - PubMed
-
- Arlt A, Vorndamm J, Muerkoster S, Yu H, Schmidt WE, Folsch UR, Schafer H. Autocrine production of interleukin 1beta confers constitutive nuclear factor kappaB activity and chemoresistance in pancreatic carcinoma cell lines. Cancer Res. 2002;62:910–916. - PubMed
-
- Bhattacharjee A, Richards WG, Staunton J, Li C, Monti S, Vasa P, Ladd C, Beheshti J, Bueno R, Gillette M, Loda M, Weber G, Mark EJ, Lander ES, Wong W, Johnson BE, Golub TR, Sugarbaker DJ, Meyerson M. Classification of human lung carcinomas by mRNA expression profiling reveals distinct adenocarcinoma subclasses. Proc Natl Acad Sci U S A. 2001;98:13790–13795. doi: 10.1073/pnas.191502998. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

