T cells that cannot respond to TGF-beta escape control by CD4(+)CD25(+) regulatory T cells
- PMID: 15753207
- PMCID: PMC2212836
- DOI: 10.1084/jem.20040685
T cells that cannot respond to TGF-beta escape control by CD4(+)CD25(+) regulatory T cells
Abstract
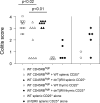
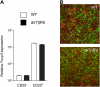
CD4(+)CD25(+) regulatory T (T reg) cells play a pivotal role in control of the immune response. Transforming growth factor-beta (TGF-beta) has been shown to be required for T reg cell activity; however, precisely how it is involved in the mechanism of suppression is poorly understood. Using the T cell transfer model of colitis, we show here that CD4(+)CD45RB(high) T cells that express a dominant negative TGF-beta receptor type II (dnTbetaRII) and therefore cannot respond to TGF-beta, escape control by T reg cells in vivo. CD4(+)CD25(+) T reg cells from the thymus of dnTbetaRII mice retain the ability to inhibit colitis, suggesting that T cell responsiveness to TGF-beta is not required for the development or peripheral function of thymic-derived T reg cells. In contrast, T reg cell activity among the peripheral dnTbetaRII CD4(+)CD25(+) population is masked by the presence of colitogenic effector cells that cannot be suppressed. Finally, we show that CD4(+)CD25(+) T reg cells develop normally in the absence of TGF-beta1 and retain the ability to suppress colitis in vivo. Importantly, the function of TGF-beta1(-/-) T reg cells was abrogated by anti-TGF-beta monoclonal antibody, indicating that functional TGF-beta can be provided by a non-T reg cell source.
Figures



References
-
- Maloy, K.J., and F. Powrie. 2001. Regulatory T cells in the control of immune pathology. Nat. Immunol. 2:816–822. - PubMed
-
- Shevach, E.M. 2002. CD4+CD25+ suppressor T cells: more questions than answers. Nat. Rev. Immunol. 2:389–400. - PubMed
-
- Takahashi, T., and S. Sakaguchi. 2003. Naturally arising CD25+CD4+ regulatory T cells in maintaining immunologic self-tolerance and preventing autoimmune disease. Curr. Mol. Med. 3:693–706. - PubMed
-
- Sakaguchi, S., N. Sakaguchi, M. Asano, M. Itoh, and M. Toda. 1995. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 155:1151–1164. - PubMed
-
- Shimizu, J., S. Yamazaki, and S. Sakaguchi. 1999. Induction of tumor immunity by removing CD25+CD4+ T cells: a common basis between tumor immunity and autoimmunity. J. Immunol. 163:5211–5218. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

