Convergent regulation of skeletal muscle Ca2+ channels by dystrophin, the actin cytoskeleton, and cAMP-dependent protein kinase
- PMID: 15753322
- PMCID: PMC554817
- DOI: 10.1073/pnas.0409695102
Convergent regulation of skeletal muscle Ca2+ channels by dystrophin, the actin cytoskeleton, and cAMP-dependent protein kinase
Abstract
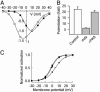
The skeletal muscle L-type Ca2+ channel (Ca(V)1.1), which is responsible for initiating muscle contraction, is regulated by phosphorylation by cAMP-dependent protein kinase (PKA) in a voltage-dependent manner that requires direct physical association between the channel and the kinase mediated through A-kinase anchoring proteins (AKAPs). The role of the actin cytoskeleton in channel regulation was investigated in skeletal myocytes cultured from wild-type mice, mdx mice that lack the cytoskeletal linkage protein dystrophin, and a skeletal muscle cell line, 129 CB3. Voltage dependence of channel activation was shifted positively, and potentiation was greatly diminished in mdx myocytes and in 129 CB3 cells treated with the microfilament stabilizer phalloidin. Voltage-dependent potentiation by strong depolarizing prepulses was reduced in mdx myocytes but could be restored by positively shifting the stimulus potentials to compensate for the positive shift in the voltage dependence of gating. Inclusion of PKA in the pipette caused a negative shift in the voltage dependence of activation and restored voltage-dependent potentiation in mdx myocytes. These results show that skeletal muscle Ca2+ channel activity and voltage-dependent potentiation are controlled by PKA and microfilaments in a convergent manner. Regulation of Ca2+ channel activity by hormones and neurotransmitters that use the PKA signal transduction pathway may interact in a critical way with the cytoskeleton and may be impaired by deletion of dystrophin, contributing to abnormal regulation of intracellular calcium concentrations in dystrophic muscle.
Figures
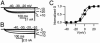
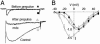
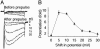
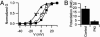
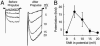
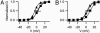
Similar articles
-
L-type Ca2+ channel function is linked to dystrophin expression in mammalian muscle.PLoS One. 2008 Mar 12;3(3):e1762. doi: 10.1371/journal.pone.0001762. PLoS One. 2008. PMID: 18516256 Free PMC article.
-
Expression of Dp260 in muscle tethers the actin cytoskeleton to the dystrophin-glycoprotein complex and partially prevents dystrophy.Hum Mol Genet. 2002 May 1;11(9):1095-105. doi: 10.1093/hmg/11.9.1095. Hum Mol Genet. 2002. PMID: 11978768
-
Involvement of TRPC in the abnormal calcium influx observed in dystrophic (mdx) mouse skeletal muscle fibers.J Cell Biol. 2002 Sep 16;158(6):1089-96. doi: 10.1083/jcb.200203091. Epub 2002 Sep 16. J Cell Biol. 2002. PMID: 12235126 Free PMC article.
-
New insights in the regulation of calcium transfers by muscle dystrophin-based cytoskeleton: implications in DMD.J Muscle Res Cell Motil. 2006;27(5-7):375-86. doi: 10.1007/s10974-006-9085-2. Epub 2006 Aug 4. J Muscle Res Cell Motil. 2006. PMID: 16897576 Review.
-
Pharmacological control of cellular calcium handling in dystrophic skeletal muscle.Neuromuscul Disord. 2002 Oct;12 Suppl 1:S155-61. doi: 10.1016/s0960-8966(02)00095-0. Neuromuscul Disord. 2002. PMID: 12206810 Review.
Cited by
-
Mutation of delta-sarcoglycan is associated with Ca(2+) -dependent vascular remodeling in the Syrian hamster.Am J Pathol. 2007 Jul;171(1):162-71. doi: 10.2353/ajpath.2007.070054. Am J Pathol. 2007. PMID: 17591963 Free PMC article.
-
Dystrophin and utrophin isoforms are expressed in glia, but not neurons, of the avian parasympathetic ciliary ganglion.Brain Res. 2008 Jul 7;1218:21-34. doi: 10.1016/j.brainres.2008.04.071. Epub 2008 May 6. Brain Res. 2008. PMID: 18533135 Free PMC article.
-
Multiple kinase pathways regulate voltage-dependent Ca2+ influx and migration in oligodendrocyte precursor cells.J Neurosci. 2010 May 5;30(18):6422-33. doi: 10.1523/JNEUROSCI.5086-09.2010. J Neurosci. 2010. PMID: 20445068 Free PMC article.
-
Identification of new dystroglycan complexes in skeletal muscle.PLoS One. 2013 Aug 8;8(8):e73224. doi: 10.1371/journal.pone.0073224. eCollection 2013. PLoS One. 2013. PMID: 23951345 Free PMC article.
-
Sites of proteolytic processing and noncovalent association of the distal C-terminal domain of CaV1.1 channels in skeletal muscle.Proc Natl Acad Sci U S A. 2005 Apr 5;102(14):5274-9. doi: 10.1073/pnas.0409885102. Epub 2005 Mar 25. Proc Natl Acad Sci U S A. 2005. PMID: 15793008 Free PMC article.
References
-
- Tanabe, T., Beam, K. G., Powell, J. A. & Numa, S. (1988) Nature 336, 134–139. - PubMed
-
- Rios, E. & Pizarro, G. (1991) Physiol. Rev. 71, 849–908. - PubMed
-
- Huerta, M., Muniz, J., Trujillo, X. & Lomeli, J. (1991) Jpn. J. Physiol. 41, 851–860. - PubMed
-
- Huang, C.-F., Flucher, B. E., Schmidt, M. M., Stroud, S. K. & Schmidt, J. (1994) Neuron 13, 167–177. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

