Enhanced fluorescence cyanide detection at physiologically lethal levels: reduced ICT-based signal transduction
- PMID: 15755185
- PMCID: PMC6844257
- DOI: 10.1021/ja044421i
Enhanced fluorescence cyanide detection at physiologically lethal levels: reduced ICT-based signal transduction
Abstract
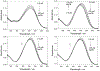
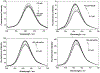
Three water-soluble fluorescent probes have been specifically designed to determine free cyanide concentrations up to physiologically lethal levels, >20 microM. The probes have been designed in such a way as to afford many notable sensing features, which render them unique with regard to signal transduction, photophysical characteristics, and their application to physiological cyanide determination and safeguard. The probes are readily able to reversibly bind free aqueous cyanide with dissociation constants around 4 microM3. Subsequent cyanide binding modulates the intramolecular charge transfer within the probes, a change in the electronic properties within the probes, resulting in enhanced fluorescence optical signals as a function of increased solution cyanide concentration. The ground-state chelation with cyanide produces wavelength shifts, which also enable the probes to sense cyanide in both an excitation and emission ratiometric manner, in addition to enhanced fluorescence signaling. This has enabled a generic cyanide sensing platform to be realized that is not dependent on fluorescent probe concentration, probe photodegradation, or fluctuations in the intensity of any employed excitation sources, ideal for remote cyanide sensing applications. Further, the >600 nm fluorescence emission of the probes potentially allows for enhanced fluorescence ratiometric cyanide sensing in the optical window of tissues and blood, facilitating their use for the transdermal monitoring of cyanide for mammalian safeguard or postmortem in fire victims, both areas of active research.
Figures

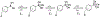


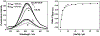


Similar articles
-
Excitation and emission wavelength ratiometric cyanide-sensitive probes for physiological sensing.Anal Biochem. 2004 Apr 1;327(1):82-90. doi: 10.1016/j.ab.2003.12.026. Anal Biochem. 2004. PMID: 15033514 Free PMC article.
-
Colorimetric probes based on anthraimidazolediones for selective sensing of fluoride and cyanide ion via intramolecular charge transfer.J Org Chem. 2011 Oct 21;76(20):8215-22. doi: 10.1021/jo201290a. Epub 2011 Sep 29. J Org Chem. 2011. PMID: 21892827
-
Ratiometric detection and monitoring of cyanide in biological, environmental and food samples by a novel triphenylamine-xhantane based fluorescent probe.Anal Chim Acta. 2024 Sep 1;1320:343000. doi: 10.1016/j.aca.2024.343000. Epub 2024 Jul 22. Anal Chim Acta. 2024. PMID: 39142780
-
Fluorescence-based ratiometric sensors as emerging tools for CN- detection: Chemical structures, sensing mechanisms and applications.Methods. 2024 Feb;222:57-80. doi: 10.1016/j.ymeth.2024.01.001. Epub 2024 Jan 6. Methods. 2024. PMID: 38191006 Review.
-
Fluorescent Probes for the Visualization of Cell Viability.Acc Chem Res. 2019 Aug 20;52(8):2147-2157. doi: 10.1021/acs.accounts.9b00289. Epub 2019 Jul 23. Acc Chem Res. 2019. PMID: 31335119 Review.
Cited by
-
A Mitochondria-Specific Fluorescent Probe for Visualizing Endogenous Hydrogen Cyanide Fluctuations in Neurons.J Am Chem Soc. 2018 Feb 7;140(5):1870-1875. doi: 10.1021/jacs.7b12545. Epub 2018 Jan 25. J Am Chem Soc. 2018. PMID: 29337546 Free PMC article.
-
Strapped calix[4]pyrroles bearing a 1,3-indanedione at a beta-pyrrolic position: chemodosimeters for the cyanide anion.Org Lett. 2009 Aug 20;11(16):3626-9. doi: 10.1021/ol901361h. Org Lett. 2009. PMID: 19639968 Free PMC article.
-
Donor-acceptor substituted phenylethynyltriphenylenes - excited state intramolecular charge transfer, solvatochromic absorption and fluorescence emission.Beilstein J Org Chem. 2010 Oct 18;6:992-1001. doi: 10.3762/bjoc.6.112. Beilstein J Org Chem. 2010. PMID: 21085512 Free PMC article.
-
A new phenothiazine-based selective visual and fluorescent sensor for cyanide.BMC Chem. 2020 Jan 7;14(1):2. doi: 10.1186/s13065-019-0656-x. eCollection 2020 Dec. BMC Chem. 2020. PMID: 31922151 Free PMC article.
-
Zinc oxide nanomaterials for biomedical fluorescence detection.J Nanosci Nanotechnol. 2014 Jan;14(1):475-86. doi: 10.1166/jnn.2014.9099. J Nanosci Nanotechnol. 2014. PMID: 24730276 Free PMC article. Review.
References
-
- Baskin SI; Brewer TG Medical Aspects of Chemical and Biological Warfare; Sidell F, Takafuji ET, Franz DR, Eds.; TMM Publications: Washington, DC, 1997; Chapter 10, pp 271–286 and references therein.
-
- Warburg O Hoppe-Seyler’s Z. Physiol. Chem 1911, 76, 331–346.
-
- Kellin D Proc. R. Soc. London, Ser. B: Biol. Sci 1929, 104, 206–251.
-
- Ishii A; Seno H; Watanabe-Suzuki K; Suzuki O; Kumazawa T Anal. Chem 1998, 70 (22), 4873–4876. - PubMed
-
- Moriva F; Hashimoto YJ For. Sci 2001, 46 (6), 1421–1425. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

