The acetate switch
- PMID: 15755952
- PMCID: PMC1082793
- DOI: 10.1128/MMBR.69.1.12-50.2005
The acetate switch
Abstract
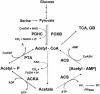
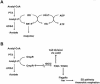
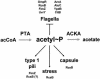
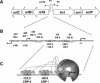
To succeed, many cells must alternate between life-styles that permit rapid growth in the presence of abundant nutrients and ones that enhance survival in the absence of those nutrients. One such change in life-style, the "acetate switch," occurs as cells deplete their environment of acetate-producing carbon sources and begin to rely on their ability to scavenge for acetate. This review explains why, when, and how cells excrete or dissimilate acetate. The central components of the "switch" (phosphotransacetylase [PTA], acetate kinase [ACK], and AMP-forming acetyl coenzyme A synthetase [AMP-ACS]) and the behavior of cells that lack these components are introduced. Acetyl phosphate (acetyl approximately P), the high-energy intermediate of acetate dissimilation, is discussed, and conditions that influence its intracellular concentration are described. Evidence is provided that acetyl approximately P influences cellular processes from organelle biogenesis to cell cycle regulation and from biofilm development to pathogenesis. The merits of each mechanism proposed to explain the interaction of acetyl approximately P with two-component signal transduction pathways are addressed. A short list of enzymes that generate acetyl approximately P by PTA-ACKA-independent mechanisms is introduced and discussed briefly. Attention is then directed to the mechanisms used by cells to "flip the switch," the induction and activation of the acetate-scavenging AMP-ACS. First, evidence is presented that nucleoid proteins orchestrate a progression of distinct nucleoprotein complexes to ensure proper transcription of its gene. Next, the way in which cells regulate AMP-ACS activity through reversible acetylation is described. Finally, the "acetate switch" as it exists in selected eubacteria, archaea, and eukaryotes, including humans, is described.
Figures







References
-
- Abdel-Hamid, A. M., M. M. Attwood, and J. R. Guest. 2001. Pyruvate oxidase contributes to the aerobic growth efficiency of Escherichia coli. Microbiology 147:1483-1498. - PubMed
-
- Allen, A. 1984. The structure and function of gastrointestinal mucus, p. 3-11. In I. E. C. Boedeker (ed.), Attachment of organisms to the gut mucosa, vol. II. CRC Press, Inc., Boca Raton, Fla.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

