Initiation of embryonic cardiac pacemaker activity by inositol 1,4,5-trisphosphate-dependent calcium signaling
- PMID: 15758029
- PMCID: PMC1087245
- DOI: 10.1091/mbc.e04-10-0883
Initiation of embryonic cardiac pacemaker activity by inositol 1,4,5-trisphosphate-dependent calcium signaling
Abstract
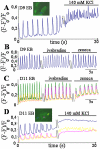
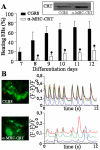
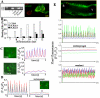
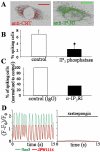
In the adult, the heart rate is driven by spontaneous and repetitive depolarizations of pacemaker cells to generate a firing of action potentials propagating along the conduction system and spreading into the ventricles. In the early embryo before E9.5, the pacemaker ionic channel responsible for the spontaneous depolarization of cells is not yet functional. Thus the mechanisms that initiate early heart rhythm during cardiogenesis are puzzling. In the absence of a functional pacemaker ionic channel, the oscillatory nature of inositol 1,4,5-trisphosphate (InsP3)-induced intracellular Ca2+ signaling could provide an alternative pacemaking mechanism. To test this hypothesis, we have engineered pacemaker cells from embryonic stem (ES) cells, a model that faithfully recapitulates early stages of heart development. We show that InsP3-dependent shuttle of free Ca2+ in and out of the endoplasmic reticulum is essential for a proper generation of pacemaker activity during early cardiogenesis and fetal life.
Figures

Similar articles
-
Calreticulin, Ca2+, and calcineurin - signaling from the endoplasmic reticulum.Mol Cells. 2004 Jun 30;17(3):383-9. Mol Cells. 2004. PMID: 15232210 Review.
-
The type 3 inositol 1,4,5-trisphosphate receptor is concentrated at the tight junction level in polarized MDCK cells.J Cell Sci. 2003 Jul 1;116(Pt 13):2791-803. doi: 10.1242/jcs.00482. Epub 2003 May 20. J Cell Sci. 2003. PMID: 12759372
-
Signal-induced Ca2+ oscillations through the regulation of the inositol 1,4,5-trisphosphate-gated Ca2+ channel: an allosteric model.J Theor Biol. 1997 Jun 7;186(3):307-26. doi: 10.1006/jtbi.1996.0365. J Theor Biol. 1997. PMID: 9219669
-
Functional interaction between InsP3 receptors and store-operated Htrp3 channels.Nature. 1998 Dec 3;396(6710):478-82. doi: 10.1038/24890. Nature. 1998. PMID: 9853757
-
Inositol 1,4,5-trisphosphate receptors in the endoplasmic reticulum: A single-channel point of view.Cell Calcium. 2015 Jul;58(1):67-78. doi: 10.1016/j.ceca.2014.12.008. Epub 2014 Dec 18. Cell Calcium. 2015. PMID: 25555684 Free PMC article. Review.
Cited by
-
Calreticulin Is Required for TGF-β-Induced Epithelial-to-Mesenchymal Transition during Cardiogenesis in Mouse Embryonic Stem Cells.Stem Cell Reports. 2017 May 9;8(5):1299-1311. doi: 10.1016/j.stemcr.2017.03.018. Epub 2017 Apr 20. Stem Cell Reports. 2017. PMID: 28434939 Free PMC article.
-
Calcium signalling in developing cardiomyocytes: implications for model systems and disease.J Physiol. 2015 Mar 1;593(5):1047-63. doi: 10.1113/jphysiol.2014.274712. Epub 2015 Feb 9. J Physiol. 2015. PMID: 25641733 Free PMC article. Review.
-
Intracellular Ca2+ oscillations, a potential pacemaking mechanism in early embryonic heart cells.J Gen Physiol. 2007 Aug;130(2):133-44. doi: 10.1085/jgp.200609575. J Gen Physiol. 2007. PMID: 17664344 Free PMC article.
-
Emerging roles of inositol 1,4,5-trisphosphate signaling in cardiac myocytes.J Mol Cell Cardiol. 2008 Aug;45(2):128-47. doi: 10.1016/j.yjmcc.2008.05.014. Epub 2008 Jun 15. J Mol Cell Cardiol. 2008. PMID: 18603259 Free PMC article. Review.
-
The expanding phenotypes of cohesinopathies: one ring to rule them all!Cell Cycle. 2019 Nov;18(21):2828-2848. doi: 10.1080/15384101.2019.1658476. Epub 2019 Sep 13. Cell Cycle. 2019. PMID: 31516082 Free PMC article. Review.
References
-
- Accili, E. A., Proenza, C., Baruscotti, M., and DiFrancesco, D. (2002). From funny current to HCN channels: 20 years of excitation. News Physiol. Sci. 17, 32-37. - PubMed
-
- Behfar, A., Zingman, L., Hodgson, D., Rauzier, J., Kane, G., Terzic, A., and Pucéat, M. (2002). Stem cell differentiation requires a paracrine pathway in the heart. FASEB J. 16, 1558-1566. - PubMed
-
- Berridge, M. J., Bootman, M. D., and Roderick, H. L. (2003). Calcium signalling: dynamics, homeostasis and remodelling. Nat. Rev. Mol. Cell. Biol. 4, 517-529. - PubMed
-
- Berridge, M. J., Lipp, P., and Bootman, M. D. (2000). The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell. Biol. 1, 11-21. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

