In vitro culture of human osteosarcoma cell lines: a comparison of functional characteristics for cell lines cultured in medium without and with fetal calf serum
- PMID: 15776274
- PMCID: PMC12161246
- DOI: 10.1007/s00432-004-0650-z
In vitro culture of human osteosarcoma cell lines: a comparison of functional characteristics for cell lines cultured in medium without and with fetal calf serum
Abstract
Purpose: Experimental in vitro models including well-characterised cell lines can be used to identify possible new therapeutic targets for the treatment of osteosarcoma. Culture media including inactivated serum is often recommended for in vitro culture of osteosarcoma cells, but the serum component then represents a nonstandardised parameter including a wide range of unidentified mediators. To improve the standardisation we have investigated whether serum-free culture media can be used in experimental in vitro studies of osteosarcoma cell lines.
Methods: The seven osteosarcoma cell lines Cal72, SJSA-1, Saos-2, SK-ES-1, U2OS, 143.98.2, and KHOS-32IH were cultured in vitro in various serum-free media and media supplemented with 10% heat-inactivated fetal calf serum (FCS).
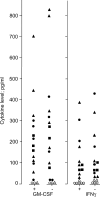
Results: Although proliferation often was relatively low in serum-free media (X-vivo 10, X-vivo 15, X-vivo 20, Stem Span SFEM), some cell lines (Cal72, KHOS-32IH, Saos-2) showed proliferation comparable with the recommended FCS-containing media even when using serum-free conditions. The optimal serum-free medium then varied between cell lines. We also compared 6 different FCS-containing media (including Stem Span with 10% FCS) and the optimal FCS-containing medium varied between cell lines. However, all cell lines proliferated well in Stem Span with FCS, and this medium was regarded as optimal for four of the lines. FCS could not be replaced by fatty acids or low density lipoprotein when testing the Stem Span medium. The release of a wide range of soluble mediators showed only minor differences when using serum-free and FCS-containing media (including Stem Span with and without FCS), and serum-free Stem Span could also be used for in vitro studies of mitogen-stimulated T cell activation in the presence of accessory osteosarcoma cells. The use of Stem Span with 10% FCS allowed the release of a wide range of chemokines by osteosarcoma cell lines (Cal72, SJSA-1), and the chemokine release profile was very similar to the fibroblast lines Hs27 and HFL1.
Conclusions: Serum-free culture media can be used for in vitro studies of several osteosarcoma cell lines, but the optimal medium varies between cell lines and thus depends on: (i) the cell lines to be investigated/compared; (ii) the functional characteristic that is evaluated (proliferation, cytokine release); and (iii) whether coculture experiments are included.
Figures

References
-
- Berge RK, Skorve J, Tronstad KJ, Berge K, Gudbrandsen OA, Grav H (2002) Metabolic effects of thia fatty acids. Curr Opin Lipidol 13:295–304 - PubMed
-
- Bruserud Ø, Gjertsen BT (2000) New strategies for the treatment of acute myelogenous leukemia: differentiation induction—present use and future possibilities. Stem Cells 18:157–165 - PubMed
-
- Bruserud Ø, Ulvestad E (2000a) Cytokine responsiveness of mitogen-activated T cells derived from acute leukemia patients with chemotherapy-induced leukopenia. J Interferon Cytokine Res 20:947–954 - PubMed
-
- Bruserud Ø, Ulvestad E (2000b) Acute myelogenous leukemia blasts as accessory cells during in vitro T lymphocyte activation. Cell Immunol 206:36–50 - PubMed
-
- Bruserud Ø, Ulvestad E, Berentsen S, Bergheim J, Nesthus I (1998) T lymphocyte functions in acute leukaemia patients with severe chemotherapy-induced cytopenia: characterisation of clonogenic T cell proliferation. Scand J Immunol 47:54–62 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

