Enhancing resistance to Sclerotinia minor in peanut by expressing a barley oxalate oxidase gene
- PMID: 15778458
- PMCID: PMC1088326
- DOI: 10.1104/pp.104.057232
Enhancing resistance to Sclerotinia minor in peanut by expressing a barley oxalate oxidase gene
Abstract
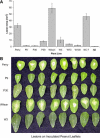
Sclerotinia minor Jagger is the causal agent of Sclerotinia blight, a highly destructive disease of peanut (Arachis hypogaea). Based on evidence that oxalic acid is involved in the pathogenicity of many Sclerotinia species, our objectives were to recover transgenic peanut plants expressing an oxalic acid-degrading oxalate oxidase and to evaluate them for increased resistance to S. minor. Transformed plants were regenerated from embryogenic cultures of three Virginia peanut cultivars (Wilson, Perry, and NC-7). A colorimetric enzyme assay was used to screen for oxalate oxidase activity in leaf tissue. Candidate plants with a range of expression levels were chosen for further analysis. Integration of the transgene was confirmed by Southern-blot analysis, and gene expression was demonstrated in transformants by northern-blot analysis. A sensitive fluorescent enzyme assay was used to quantify expression levels for comparison to the colorimetric protocol. A detached leaflet assay tested whether transgene expression could limit lesion size resulting from direct application of oxalic acid. Lesion size was significantly reduced in transgenic plants compared to nontransformed controls (65%-89% reduction at high oxalic acid concentrations). A second bioassay examined lesion size after inoculation of leaflets with S. minor mycelia. Lesion size was reduced by 75% to 97% in transformed plants, providing evidence that oxalate oxidase can confer enhanced resistance to Sclerotinia blight in peanut.
Figures
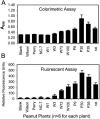
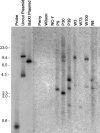
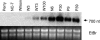

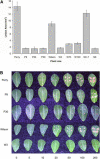
Similar articles
-
Sclerotinia blight resistance in Virginia-type peanut transformed with a barley oxalate oxidase gene.Phytopathology. 2011 Jul;101(7):786-93. doi: 10.1094/PHYTO-10-10-0266. Phytopathology. 2011. PMID: 21303213
-
Enhanced resistance to sclerotinia stem rot in transgenic soybean that overexpresses a wheat oxalate oxidase.Transgenic Res. 2019 Feb;28(1):103-114. doi: 10.1007/s11248-018-0106-x. Epub 2018 Nov 26. Transgenic Res. 2019. PMID: 30478526
-
Assessment of peanut quality and compositional characteristics among transgenic sclerotinia blight-resistant and non-transgenic susceptible cultivars.J Agric Food Chem. 2014 Aug 6;62(31):7877-85. doi: 10.1021/jf5016297. Epub 2014 Jul 22. J Agric Food Chem. 2014. PMID: 24972023
-
Sclerotinia sclerotiorum: An Evaluation of Virulence Theories.Annu Rev Phytopathol. 2018 Aug 25;56:311-338. doi: 10.1146/annurev-phyto-080417-050052. Epub 2018 Jun 29. Annu Rev Phytopathol. 2018. PMID: 29958073 Review.
-
Manipulation of oxalate metabolism in plants for improving food quality and productivity.Phytochemistry. 2019 Feb;158:103-109. doi: 10.1016/j.phytochem.2018.10.029. Epub 2018 Nov 28. Phytochemistry. 2019. PMID: 30500595 Review.
Cited by
-
Nickel deficiency disrupts metabolism of ureides, amino acids, and organic acids of young pecan foliage.Plant Physiol. 2006 Feb;140(2):433-43. doi: 10.1104/pp.105.072983. Epub 2006 Jan 13. Plant Physiol. 2006. PMID: 16415214 Free PMC article.
-
Identification and analysis of the germin-like gene family in soybean.BMC Genomics. 2010 Nov 8;11:620. doi: 10.1186/1471-2164-11-620. BMC Genomics. 2010. PMID: 21059215 Free PMC article.
-
Transcriptomic Analysis of Quinoa Reveals a Group of Germin-Like Proteins Induced by Trichoderma.Front Fungal Biol. 2021 Dec 1;2:768648. doi: 10.3389/ffunb.2021.768648. eCollection 2021. Front Fungal Biol. 2021. PMID: 37744129 Free PMC article.
-
Progress in genetic engineering and genome editing of peanuts: revealing the future of crop improvement.Physiol Mol Biol Plants. 2024 Nov;30(11):1759-1775. doi: 10.1007/s12298-024-01534-6. Epub 2024 Dec 2. Physiol Mol Biol Plants. 2024. PMID: 39687700 Review.
-
Identification of seed proteins associated with resistance to pre-harvested aflatoxin contamination in peanut (Arachis hypogaea L).BMC Plant Biol. 2010 Nov 30;10:267. doi: 10.1186/1471-2229-10-267. BMC Plant Biol. 2010. PMID: 21118527 Free PMC article.
References
-
- Bateman DF, Beer SV (1965) Simultaneous production and synergistic action of oxalic acid and polygalacturonase during pathogenesis by Sclerotium rolfsii. Phytopathology 55: 204–211 - PubMed
-
- Bennett AR, Hindal DF (1989) Mycelial growth and oxalate production by five strains of Cryphonectria parasitica in selected liquid culture media. Mycologia 81: 554–560
-
- Berna A, Bernier F (1999) Regulation by biotic and abiotic stress of a wheat germin gene encoding oxalate oxidase, a H2O2-producing enzyme. Plant Mol Biol 39: 539–549 - PubMed
-
- Chenault KD, Burns JA, Melouk HA, Payton ME (2002) Hydrolase activity in transgenic peanut. Peanut Sci 29: 89–95
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

