Defining the role of active-site loop fluctuations in dihydrofolate reductase catalysis
- PMID: 15795383
- PMCID: PMC556001
- DOI: 10.1073/pnas.0500699102
Defining the role of active-site loop fluctuations in dihydrofolate reductase catalysis
Abstract
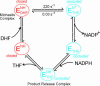
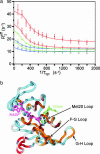
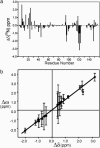
Dynamic processes are implicit in the catalytic function of all enzymes. To obtain insights into the relationship between the dynamics and thermodynamics of protein fluctuations and catalysis, we have measured millisecond time scale motions in the enzyme dihydrofolate reductase using NMR relaxation methods. Studies of a ternary complex formed from the substrate analog folate and oxidized NADP+ cofactor revealed conformational exchange between a ground state, in which the active site loops adopt a closed conformation, and a weakly populated (4.2% at 30 degrees C) excited state with the loops in the occluded conformation. Fluctuations between these states, which involve motions of the nicotinamide ring of the cofactor into and out of the active site, occur on a time scale that is directly relevant to the structural transitions involved in progression through the catalytic cycle.
Figures

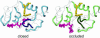
References
-
- Careri, G., Fasella, P. & Gratton, E. (1979) Annu. Rev. Biophys. Bioeng. 8, 69–97. - PubMed
-
- Gavish, B. & Werber, M. M. (1979) Biochemistry 18, 1269–1275. - PubMed
-
- Cannon, W. R., Singleton, S. F. & Benkovic, S. J. (1996) Nat. Struct. Biol. 3, 821–833. - PubMed
-
- Osborne, M. J., Schnell, J., Benkovic, S. J., Dyson, H. J. & Wright, P. E. (2001) Biochemistry 40, 9846–9859. - PubMed
-
- Eisenmesser, E. Z., Bosco, D. A., Akke, M. & Kern, D. (2002) Science 295, 1520–1523. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

