Amantadine inhibits NMDA receptors by accelerating channel closure during channel block
- PMID: 15800186
- PMCID: PMC6724906
- DOI: 10.1523/JNEUROSCI.4262-04.2005
Amantadine inhibits NMDA receptors by accelerating channel closure during channel block
Abstract
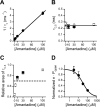
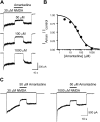
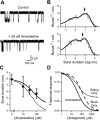
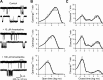
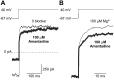
The channel of NMDA receptors is blocked by a wide variety of drugs. NMDA receptor channel blockers include drugs of abuse that induce psychotic behavior, such as phencyclidine, and drugs with wide therapeutic utility, such as amantadine and memantine. We describe here the molecular mechanism of amantadine inhibition. In contrast to most other described channel-blocking molecules, amantadine causes the channel gate of NMDA receptors to close more quickly. Our results confirm that amantadine binding inhibits current flow through NMDA receptor channels but show that its main inhibitory action at pharmaceutically relevant concentrations results from stabilization of closed states of the channel. The surprising variation in the clinical utility of NMDA channel blockers may in part derive from their diverse effects on channel gating.
Figures


References
-
- Antonov SM, Gmiro VE, Johnson JW (1998) Binding sites for permeant ions in the channel of NMDA receptors and their effects on channel block. Nat Neurosci 1: 451-461. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
