Relationship between membrane phosphatidylinositol-4,5-bisphosphate and receptor-mediated inhibition of native neuronal M channels
- PMID: 15800195
- PMCID: PMC6724893
- DOI: 10.1523/JNEUROSCI.3231-04.2005
Relationship between membrane phosphatidylinositol-4,5-bisphosphate and receptor-mediated inhibition of native neuronal M channels
Abstract
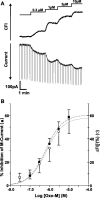
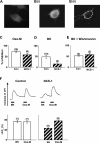
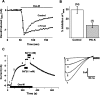
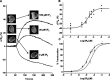
The relationship between receptor-induced membrane phosphatidylinositol-4'5'-bisphosphate (PIP2) hydrolysis and M-current inhibition was assessed in single-dissociated rat sympathetic neurons by simultaneous or parallel recording of membrane current and membrane-to-cytosol translocation of the fluorescent PIP2/inositol 1,4,5-trisphosphate (IP3)-binding peptide green fluorescent protein-tagged pleckstrin homology domain of phospholipase C (GFP-PLCdelta-PH). The muscarinic receptor agonist oxotremorine-M produced parallel time- and concentration-dependent M-current inhibition and GFP-PLCdelta-PH translocation; bradykinin also produced parallel time-dependent inhibition and translocation. Phosphatidylinositol-4-phosphate-5-kinase (PI5-K) overexpression reduced both M-current inhibition and GFP-PLCdelta-PH translocation by both oxotremorine-M and bradykinin. These effects were partly reversed by wortmannin, which inhibits phosphatidylinositol-4-kinase (PI4-K). PI5-K overexpression also reduced the inhibitory action of oxotremorine-M on PIP2-gated G-protein-gated inward rectifier (Kir3.1/3.2) channels; bradykinin did not inhibit these channels. Overexpression of neuronal calcium sensor-1 protein (NCS-1), which increases PI4-K activity, did not affect responses to oxotremorine-M but reduced both fluorescence translocation and M-current inhibition by bradykinin. Using an intracellular IP3 membrane fluorescence-displacement assay, initial mean concentrations of membrane [PIP2] were estimated at 261 microm (95% confidence limit; 192-381 microm), rising to 693 microm (417-1153 microm) in neurons overexpressing PI5-K. Changes in membrane [PIP2] during application of oxotremorine-M were calculated from fluorescence data. The results, taken in conjunction with previous data for KCNQ2/3 (Kv7.2/Kv7.3) channel gating by PIP2 (Zhang et al., 2003), accorded with the hypothesis that the inhibitory action of oxotremorine-M on M current resulted from depletion of PIP2. The effects of bradykinin require additional components of action, which might involve IP3-induced Ca2+ release and consequent M-channel inhibition (as proposed previously) and stimulation of PIP2 synthesis by Ca2+-dependent activation of NCS-1.
Figures




Similar articles
-
Phosphatidylinositol [correction] 4,5-bisphosphate signals underlie receptor-specific Gq/11-mediated modulation of N-type Ca2+ channels.J Neurosci. 2004 Dec 1;24(48):10980-92. doi: 10.1523/JNEUROSCI.3869-04.2004. J Neurosci. 2004. PMID: 15574748 Free PMC article.
-
Modulation of Gq-protein-coupled inositol trisphosphate and Ca2+ signaling by the membrane potential.J Neurosci. 2006 Sep 27;26(39):9983-95. doi: 10.1523/JNEUROSCI.2773-06.2006. J Neurosci. 2006. PMID: 17005862 Free PMC article.
-
Regulation of Kv7 (KCNQ) K+ channel open probability by phosphatidylinositol 4,5-bisphosphate.J Neurosci. 2005 Oct 26;25(43):9825-35. doi: 10.1523/JNEUROSCI.2597-05.2005. J Neurosci. 2005. PMID: 16251430 Free PMC article.
-
Neural KCNQ (Kv7) channels.Br J Pharmacol. 2009 Apr;156(8):1185-95. doi: 10.1111/j.1476-5381.2009.00111.x. Epub 2009 Mar 9. Br J Pharmacol. 2009. PMID: 19298256 Free PMC article. Review.
-
Channelopathies linked to plasma membrane phosphoinositides.Pflugers Arch. 2010 Jul;460(2):321-41. doi: 10.1007/s00424-010-0828-y. Epub 2010 Apr 16. Pflugers Arch. 2010. PMID: 20396900 Free PMC article. Review.
Cited by
-
Coordinated signal integration at the M-type potassium channel upon muscarinic stimulation.EMBO J. 2012 May 29;31(14):3147-56. doi: 10.1038/emboj.2012.156. EMBO J. 2012. PMID: 22643219 Free PMC article.
-
Regulation of Ca(V)2 calcium channels by G protein coupled receptors.Biochim Biophys Acta. 2013 Jul;1828(7):1629-43. doi: 10.1016/j.bbamem.2012.10.004. Epub 2012 Oct 12. Biochim Biophys Acta. 2013. PMID: 23063655 Free PMC article. Review.
-
Modulation of cyclic nucleotide-regulated HCN channels by PIP(2) and receptors coupled to phospholipase C.Pflugers Arch. 2007 Oct;455(1):125-45. doi: 10.1007/s00424-007-0295-2. Epub 2007 Jun 29. Pflugers Arch. 2007. PMID: 17605039
-
An M2-like muscarinic receptor enhances a delayed rectifier K+ current in rat sympathetic neurones.Br J Pharmacol. 2006 Oct;149(4):441-9. doi: 10.1038/sj.bjp.0706874. Epub 2006 Sep 4. Br J Pharmacol. 2006. PMID: 16953191 Free PMC article.
-
Receptor-specific inhibition of GABAB-activated K+ currents by muscarinic and metabotropic glutamate receptors in immature rat hippocampus.J Physiol. 2007 Apr 15;580(Pt. 2):411-22. doi: 10.1113/jphysiol.2006.125914. Epub 2007 Jan 25. J Physiol. 2007. PMID: 17255165 Free PMC article.
References
-
- Agard DA, Hiraoka Y, Shaw P, Sedat JW (1989) Fluorescence microscopy in three dimensions. Methods Cell Biol 30: 353-377. - PubMed
-
- Bender K, Wellner-Kienitz M-C, Pott L (2002) Transfection of a phosphatidyl-4-phosphate 5-kinase gene into rat atrial myocytes removes inhibition of GIRK current by endothelin and α-adrenergic agonists. FEBS Lett 529: 356-360. - PubMed
-
- Bofill-Cardona E, Vartian N, Nanoff C, Freissmuth M, Boehm S (2000) Two different signaling mechanisms involved in the excitation of rat sympathetic neurons by uridine nucleotides. Mol Pharmacol 57: 1165-1172. - PubMed
-
- Brown DA (1988) M-currents. In: Ion channels, Vol 1 (Narahashi T, ed), pp 55-99.New York: Plenum. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
