AMPA/kainate receptor-mediated downregulation of GABAergic synaptic transmission by calcineurin after seizures in the developing rat brain
- PMID: 15800199
- PMCID: PMC6724904
- DOI: 10.1523/JNEUROSCI.0204-05.2005
AMPA/kainate receptor-mediated downregulation of GABAergic synaptic transmission by calcineurin after seizures in the developing rat brain
Abstract
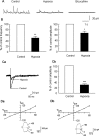
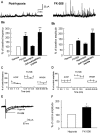
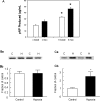
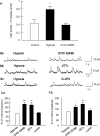
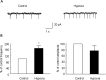
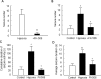
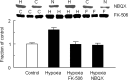
Hypoxia is the most common cause of perinatal seizures and can be refractory to conventional anticonvulsant drugs, suggesting an age-specific form of epileptogenesis. A model of hypoxia-induced seizures in immature rats reveals that seizures result in immediate activation of the phosphatase calcineurin (CaN) in area CA1 of hippocampus. After seizures, CA1 pyramidal neurons exhibit a downregulation of GABA(A) receptor (GABA(A)R)-mediated inhibition that was reversed by CaN inhibitors. CaN activation appears to be dependent on seizure-induced activation of Ca2+-permeable AMPA receptors (AMPARs), because the upregulation of CaN activation and GABA(A)R inhibition were attenuated by GYKI 52466 [1-(4-aminophenyl)-4-methyl-7,8-methylenedioxy-5H-2,3-benzodiazepine hydrochloride] or Joro spider toxin. GABA(A)R beta2/3 subunit protein was dephosphorylated at 1 h after seizures, suggesting this subunit as a possible substrate of CaN in this model. Finally, in vivo administration of the CaN inhibitor FK-506 significantly suppressed hypoxic seizures, and posttreatment with NBQX (2,3-dihydroxy-6-nitro-7-sulfonyl-benzo[f]quinoxaline) or FK-506 blocked the hypoxic seizure-induced increase in CaN expression. These data suggest that Ca2+-permeable AMPARs and CaN regulate inhibitory synaptic transmission in a novel plasticity pathway that may play a role in epileptogenesis in the immature brain.
Figures
Comment in
-
Hypoxia results in GABAergic channelopathy.Epilepsy Curr. 2005 Nov-Dec;5(6):234-5. doi: 10.1111/j.1535-7511.2005.00073.x. Epilepsy Curr. 2005. PMID: 16372059 Free PMC article. No abstract available.
Similar articles
-
Late embryonic expression of AMPA receptor function in the CA1 region of the intact hippocampus in vitro.Eur J Neurosci. 1999 Nov;11(11):4015-23. doi: 10.1046/j.1460-9568.1999.00831.x. Eur J Neurosci. 1999. PMID: 10583490
-
Postsynaptic IP3 receptor-mediated Ca2+ release modulates synaptic transmission in hippocampal neurons.Brain Res Mol Brain Res. 2005 Apr 27;135(1-2):232-48. doi: 10.1016/j.molbrainres.2004.12.018. Brain Res Mol Brain Res. 2005. PMID: 15857686
-
Inhibitory synapses in the developing auditory system are glutamatergic.Nat Neurosci. 2005 Mar;8(3):332-8. doi: 10.1038/nn1397. Epub 2005 Jan 30. Nat Neurosci. 2005. PMID: 15746915
-
Seizures beget seizures: the quest for GABA as a key player.Crit Rev Neurobiol. 2006;18(1-2):135-44. doi: 10.1615/critrevneurobiol.v18.i1-2.140. Crit Rev Neurobiol. 2006. PMID: 17725516 Review.
-
Double-edged GABAergic synaptic transmission in seizures: The importance of chloride plasticity.Brain Res. 2018 Dec 15;1701:126-136. doi: 10.1016/j.brainres.2018.09.008. Epub 2018 Sep 7. Brain Res. 2018. PMID: 30201259 Review.
Cited by
-
Decreased A-currents in hippocampal dentate granule cells after seizure-inducing hypoxia in the immature rat.Epilepsia. 2013 Jul;54(7):1223-31. doi: 10.1111/epi.12150. Epub 2013 Apr 5. Epilepsia. 2013. PMID: 23815572 Free PMC article.
-
The effects of early-life seizures on hippocampal dendrite development and later-life learning and memory.Brain Res Bull. 2014 Apr;103:39-48. doi: 10.1016/j.brainresbull.2013.10.004. Epub 2013 Oct 15. Brain Res Bull. 2014. PMID: 24140049 Free PMC article. Review.
-
New drug classes for the treatment of partial onset epilepsy: focus on perampanel.Ther Clin Risk Manag. 2013;9:285-93. doi: 10.2147/TCRM.S37317. Epub 2013 Jul 8. Ther Clin Risk Manag. 2013. PMID: 23874099 Free PMC article.
-
Chronic benzodiazepine-induced reduction in GABA(A) receptor-mediated synaptic currents in hippocampal CA1 pyramidal neurons prevented by prior nimodipine injection.Neuroscience. 2008 Nov 11;157(1):153-63. doi: 10.1016/j.neuroscience.2008.08.049. Epub 2008 Aug 27. Neuroscience. 2008. PMID: 18805463 Free PMC article.
-
Calcium-permeable AMPA receptors are expressed in a rodent model of status epilepticus.Ann Neurol. 2012 Jul;72(1):91-102. doi: 10.1002/ana.23570. Ann Neurol. 2012. PMID: 22829271 Free PMC article.
References
-
- Bergamasco B, Penna P, Ferrero P, Gavinelli R (1984) Neonatal hypoxia and epileptic risk: a clinical prospective study. Epilepsia 25: 131-146. - PubMed
-
- Bernes SM, Kaplan AM (1994) Evolution of neonatal seizures. Pediatr Clin North Am 41: 1069-1104. - PubMed
-
- Blair RE, Churn SB, Sombati S, Lou JK, DeLorenzo RJ (1999) Long-lasting decrease in neuronal Ca2+/calmodulin-dependent protein kinase II activity in a hippocampal neuronal culture model of spontaneous recurrent seizures. Brain Res 851: 54-65. - PubMed
-
- Brandon NJ, Delmas P, Kittler JT, McDonald BJ, Sieghart W, Brown DA, Smart TG, Moss SJ (2000) GABAA receptor phosphorylation and functional modulation in cortical neurons by a protein kinase C-dependent pathway. J Biol Chem 275: 38856-38862. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
