Absence of circadian variation in the pharmacokinetics of lopinavir/ritonavir given as a once daily dosing regimen in HIV-1-infected patients
- PMID: 15801934
- PMCID: PMC1884801
- DOI: 10.1111/j.1365-2125.2005.02337.x
Absence of circadian variation in the pharmacokinetics of lopinavir/ritonavir given as a once daily dosing regimen in HIV-1-infected patients
Abstract
Aims: To compare the pharmacokinetics of lopinavir/ritonavir (LPV/r) 800/200 mg administered once daily in the morning compared with the evening.
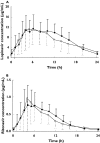
Methods: This was a randomized, two-way, cross-over study in HIV+ subjects. In each subject the pharmacokinetics of each drug were characterized after 2 weeks of LPV/r 800/200 mg administered once daily at 08.00 h and 19.00 h. On study days, LPV/r was taken with a standardized meal (800 kCal, 25% from fat) after fasting for at least 5 h. LPV/r concentrations were measured by LC-MS/MS, and the data were analyzed by noncompartmental pharmacokinetic analysis.
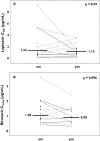
Results: Fourteen subjects completed the study (all men, mean age/weight 44 year/81 kg). The median (interquartile range) LPV AUC(0,24 h), maximum plasma concentration (C(max)) and concentration at the end of the dosing interval (C(24 h)) after am and pm dosing was, respectively, 143 (116-214) mg l(-1) h, 12.8 (10.3-17.2) mg l(-1), 1.34 (0.58-3.25) mg l(-1), and 171 (120-232) mg l(-1) h, 12.9 (8.22-16.3) mg l(-1), 1.15 (0.59-1.98) mg l(-1). The geometric mean ratio (GMR, am : pm) and 95% CI of the LPV AUC(0,24 h), C(max), and C(24 h) was 0.91 (0.79, 1.06), 1.11 (0.94, 1.32), and 1.19 (0.72, 1.96), respectively. The median ritonavir C(max) after am and pm dosing was 1.05 and 0.90 mg l(-1), respectively. The GMR (95% CI) of the RTV AUC(0,24 h), C(max), and C(24 h) was 0.93 (0.80, 1.08), 1.27 (1.00, 1.63), and 1.04 (0.68, 1.60), respectively. Administration of LPV/r in a once-daily regimen was generally well tolerated.
Conclusions: No differences were observed in the pharmacokinetics of LPV/r after am or pm dosing with food, which suggests that this once daily combination, can be taken in the morning or evening. Such flexibility in dosing may improve adherence.
Figures
Similar articles
-
Pharmacokinetics and tolerability of once- versus twice-daily lopinavir/ritonavir treatment in HIV-1-infected children.Antivir Ther. 2009;14(4):603-6. Antivir Ther. 2009. PMID: 19578247 Clinical Trial.
-
Pharmacokinetics of a once-daily regimen of lopinavir/ritonavir in HIV-1-infected children.Antivir Ther. 2006;11(4):439-45. Antivir Ther. 2006. PMID: 16856617 Clinical Trial.
-
Reduced lopinavir exposure during pregnancy.AIDS. 2006 Oct 3;20(15):1931-9. doi: 10.1097/01.aids.0000247114.43714.90. AIDS. 2006. PMID: 16988514 Clinical Trial.
-
Prediction of area under the concentration-time curve for lopinavir from peak or trough lopinavir concentrations in patients receiving lopinavir-ritonavir therapy.Am J Health Syst Pharm. 2016 Mar 15;73(6):376-85. doi: 10.2146/ajhp150417. Am J Health Syst Pharm. 2016. PMID: 26953282 Review.
-
[Penetration in reservoirs].Enferm Infecc Microbiol Clin. 2008 Dec;26 Suppl 16:41-6. doi: 10.1016/s0213-005x(08)76610-2. Enferm Infecc Microbiol Clin. 2008. PMID: 19572444 Review. Spanish.
Cited by
-
Simultaneous population pharmacokinetic model for lopinavir and ritonavir in HIV-infected adults.Clin Pharmacokinet. 2008;47(10):681-92. doi: 10.2165/00003088-200847100-00005. Clin Pharmacokinet. 2008. PMID: 18783298 Clinical Trial.
-
Adherence to anti retroviral therapy (ART) during Muslim Ramadan fasting.AIDS Behav. 2009 Feb;13(1):42-5. doi: 10.1007/s10461-008-9412-2. Epub 2008 Jun 3. AIDS Behav. 2009. PMID: 18521736 Free PMC article.
-
Model-based approach to dose optimization of lopinavir/ritonavir when co-administered with rifampicin.Br J Clin Pharmacol. 2012 May;73(5):758-67. doi: 10.1111/j.1365-2125.2011.04154.x. Br J Clin Pharmacol. 2012. PMID: 22126409 Free PMC article.
-
Relationship between HIV protease inhibitors and QTc interval duration in HIV-infected patients: a cross-sectional study.Br J Clin Pharmacol. 2009 Jan;67(1):76-82. doi: 10.1111/j.1365-2125.2008.03332.x. Epub 2008 Nov 17. Br J Clin Pharmacol. 2009. PMID: 19076152 Free PMC article.
-
Abacavir Exposure in Children Cotreated for Tuberculosis with Rifampin and Superboosted Lopinavir-Ritonavir.Antimicrob Agents Chemother. 2020 Apr 21;64(5):e01923-19. doi: 10.1128/AAC.01923-19. Print 2020 Apr 21. Antimicrob Agents Chemother. 2020. PMID: 32071055 Free PMC article.
References
-
- Bruguerolle B. Chronopharmacokinetics. Current status. Clin Pharmacokinet. 1998;35:83–94. - PubMed
-
- Bortolotti Annese V, Coccia G. Twenty-four hour ambulatory antroduodenal manometry in normal subjects (co-operative study) Neurogastroenterol Mot. 2000;12:231–8. - PubMed
-
- Ohno M, Yamaguchi I, Ito T, Saiki K, Yammamoto I, Zuma J. Circadian variation of the urinary 6β-hydroxycortisol to cortisol ratio that would reflect hepatic CYP3A4 activity. Eur J Clin Pharmacokinet. 2000;55:861–5. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

