The ATP-dependent membrane localization of protein kinase Calpha is regulated by Ca2+ influx and phosphatidylinositol 4,5-bisphosphate in differentiated PC12 cells
- PMID: 15814842
- PMCID: PMC1142429
- DOI: 10.1091/mbc.e05-01-0067
The ATP-dependent membrane localization of protein kinase Calpha is regulated by Ca2+ influx and phosphatidylinositol 4,5-bisphosphate in differentiated PC12 cells
Abstract
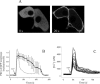
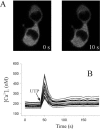
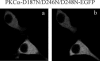
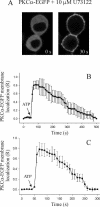
Signal transduction through protein kinase Cs (PKCs) strongly depends on their subcellular localization. Here, we investigate the molecular determinants of PKCalpha localization by using a model system of neural growth factor (NGF)-differentiated pheochromocytoma (PC12) cells and extracellular stimulation with ATP. Strikingly, the Ca2+ influx, initiated by the ATP stimulation of P2X receptors, rather than the Ca2+ released from the intracellular stores, was the driving force behind the translocation of PKCalpha to the plasma membrane. Furthermore, the localization process depended on two regions of the C2 domain: the Ca2+-binding region and the lysine-rich cluster, which bind Ca2+ and phosphatidylinositol 4,5-bisphosphate [PtdIns(4,5)P2], respectively. It was demonstrated that diacylglycerol was not involved in the localization of PKCalpha through its C1 domain, and in lieu, the presence of PtdIns(4,5)P2 increased the permanence of PKCalpha in the plasma membrane. Finally, it also was shown that ATP cooperated with NGF during the differentiation process of PC12 cells by increasing the length of the neurites, an effect that was inhibited when the cells were incubated in the presence of a specific inhibitor of PKCalpha, suggesting a possible role for this isoenzyme in the neural differentiation process. Overall, these results show a novel mechanism of PKCalpha activation in differentiated PC12 cells, where Ca2+ influx, together with the endogenous PtdIns(4,5)P2, anchor PKCalpha to the plasma membrane through two distinct motifs of its C2 domain, leading to enzyme activation.
Figures






Similar articles
-
The PtdIns(4,5)P2 ligand itself influences the localization of PKCalpha in the plasma membrane of intact living cells.J Mol Biol. 2008 Apr 4;377(4):1038-52. doi: 10.1016/j.jmb.2007.12.011. Epub 2007 Dec 8. J Mol Biol. 2008. PMID: 18304574
-
The C2 domain of PKCalpha is a Ca2+ -dependent PtdIns(4,5)P2 sensing domain: a new insight into an old pathway.J Mol Biol. 2006 Oct 6;362(5):901-14. doi: 10.1016/j.jmb.2006.07.093. Epub 2006 Aug 9. J Mol Biol. 2006. PMID: 16949603
-
The C2 domains of classical PKCs are specific PtdIns(4,5)P2-sensing domains with different affinities for membrane binding.J Mol Biol. 2007 Aug 17;371(3):608-21. doi: 10.1016/j.jmb.2007.05.086. Epub 2007 Jun 2. J Mol Biol. 2007. PMID: 17586528
-
The C2 domains of classical and novel PKCs as versatile decoders of membrane signals.Biofactors. 2010 Jan-Feb;36(1):1-7. doi: 10.1002/biof.68. Biofactors. 2010. PMID: 20049899 Review.
-
Localization of phosphatidylinositol 4,5-P(2) important in exocytosis and a quantitative analysis of chromaffin granule motion adjacent to the plasma membrane.Ann N Y Acad Sci. 2002 Oct;971:232-43. doi: 10.1111/j.1749-6632.2002.tb04467.x. Ann N Y Acad Sci. 2002. PMID: 12438123 Review.
Cited by
-
Membrane-surface anchoring of charged diacylglycerol-lactones correlates with biological activities.Chembiochem. 2010 Sep 24;11(14):2003-9. doi: 10.1002/cbic.201000343. Chembiochem. 2010. PMID: 20715268 Free PMC article.
-
Specific translocation of protein kinase Calpha to the plasma membrane requires both Ca2+ and PIP2 recognition by its C2 domain.Mol Biol Cell. 2006 Jan;17(1):56-66. doi: 10.1091/mbc.e05-06-0499. Epub 2005 Oct 19. Mol Biol Cell. 2006. PMID: 16236797 Free PMC article.
-
Classical protein kinases C are regulated by concerted interaction with lipids: the importance of phosphatidylinositol-4,5-bisphosphate.Biophys Rev. 2014 Mar;6(1):3-14. doi: 10.1007/s12551-013-0125-z. Epub 2013 Nov 27. Biophys Rev. 2014. PMID: 28509956 Free PMC article. Review.
-
The annexins: spatial and temporal coordination of signaling events during cellular stress.Cell Mol Life Sci. 2009 Aug;66(16):2623-42. doi: 10.1007/s00018-009-0027-1. Epub 2009 Apr 21. Cell Mol Life Sci. 2009. PMID: 19381436 Free PMC article. Review.
-
Oxidative stress mediates the disruption of airway epithelial tight junctions through a TRPM2-PLCγ1-PKCα signaling pathway.Int J Mol Sci. 2013 Apr 29;14(5):9475-86. doi: 10.3390/ijms14059475. Int J Mol Sci. 2013. PMID: 23629676 Free PMC article.
References
-
- Aderen, A. (1995). The MARCKS family of protein kinase C substrates. Biochem. Soc. Trans. 23, 587–591. - PubMed
-
- Bai, J., Tucker, W. C., and Chapman, E. R. (2004). PtdIns(4,5)P2 increases the speed of response of synaptotagmin and steers its membrane-penetration activity towards the plasma membrane. Nat. Struct. Mol. Biol. 11, 36–44. - PubMed
-
- Bolsover, S., Gomez-Fernandez, J. C., and Corbalán-García, S. (2003). Role of the Ca2+/phosphatidylserine binding region of the C2 domain in the translocation of protein kinase Cα to the plasma membrane. J. Biol. Chem. 278, 10282–102890. - PubMed
-
- Conesa-Zamora, P., Lopez-Andreo, M. J., Gómez-Fernández, J. C., and Corbalán-García, S. (2001). Identification of the phosphatidylserine binding site in the C2 domain that is important for protein kinase Cα activation and in vivo cell localization. Biochemistry 40, 13898–13905. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

