Hemodynamic profile, responsiveness to anandamide, and baroreflex sensitivity of mice lacking fatty acid amide hydrolase
- PMID: 15821037
- PMCID: PMC2225481
- DOI: 10.1152/ajpheart.00107.2005
Hemodynamic profile, responsiveness to anandamide, and baroreflex sensitivity of mice lacking fatty acid amide hydrolase
Abstract
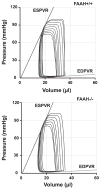
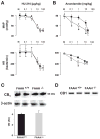
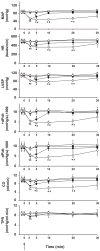
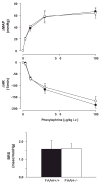
The endocannabinoid anandamide exerts neurobehavioral, cardiovascular, and immune-regulatory effects through cannabinoid receptors (CB). Fatty acid amide hydrolase (FAAH) is an enzyme responsible for the in vivo degradation of anandamide. Recent experimental studies have suggested that targeting the endocannabinergic system by FAAH inhibitors is a promising novel approach for the treatment of anxiety, inflammation, and hypertension. In this study, we compared the cardiac performance of FAAH knockout (FAAH-/-) mice and their wild-type (FAAH+/+) littermates and analyzed the hemodynamic effects of anandamide using the Millar pressure-volume conductance catheter system. Baseline cardiovascular parameters, systolic and diastolic function at different preloads, and baroreflex sensitivity were similar in FAAH-/- and FAAH+/+ mice. FAAH-/- mice displayed increased sensitivity to anandamide-induced, CB1-mediated hypotension and decreased cardiac contractility compared with FAAH(+/+) littermates. In contrast, the hypotensive potency of synthetic CB1 agonist HU-210 and the level of expression of myocardial CB1 were similar in the two strains. The myocardial levels of anandamide and oleoylethanolamide, but not 2-arachidonylglycerol, were increased in FAAH-/- mice compared with FAAH+/+ mice. These results indicate that mice lacking FAAH have a normal hemodynamic profile, and their increased responsiveness to anandamide-induced hypotension and cardiodepression is due to the decreased degradation of anandamide rather than an increase in target organ sensitivity to CB1 agonists.
Figures


Comment in
-
Is there a role for anandamide in cardiovascular regulation? Insights from studies of endocannabinoid metabolism.Am J Physiol Heart Circ Physiol. 2005 Aug;289(2):H520-1. doi: 10.1152/ajpheart.00433.2005. Am J Physiol Heart Circ Physiol. 2005. PMID: 16014613 Review. No abstract available.
Similar articles
-
Assessment of anandamide's pharmacological effects in mice deficient of both fatty acid amide hydrolase and cannabinoid CB1 receptors.Eur J Pharmacol. 2007 Feb 14;557(1):44-8. doi: 10.1016/j.ejphar.2006.11.002. Epub 2006 Nov 10. Eur J Pharmacol. 2007. PMID: 17217945
-
Phenotypic assessment of THC discriminative stimulus properties in fatty acid amide hydrolase knockout and wildtype mice.Neuropharmacology. 2015 Jun;93:237-42. doi: 10.1016/j.neuropharm.2015.02.004. Epub 2015 Feb 16. Neuropharmacology. 2015. PMID: 25698527 Free PMC article.
-
Haemodynamic profile and responsiveness to anandamide of TRPV1 receptor knock-out mice.J Physiol. 2004 Jul 15;558(Pt 2):647-57. doi: 10.1113/jphysiol.2004.064824. Epub 2004 Apr 30. J Physiol. 2004. PMID: 15121805 Free PMC article.
-
Fatty acid amide hydrolase: an emerging therapeutic target in the endocannabinoid system.Curr Opin Chem Biol. 2003 Aug;7(4):469-75. doi: 10.1016/s1367-5931(03)00079-6. Curr Opin Chem Biol. 2003. PMID: 12941421 Review.
-
Oxidation of the endogenous cannabinoid arachidonoyl ethanolamide by the cytochrome P450 monooxygenases: physiological and pharmacological implications.Pharmacol Rev. 2010 Mar;62(1):136-54. doi: 10.1124/pr.109.001081. Epub 2010 Feb 4. Pharmacol Rev. 2010. PMID: 20133390 Free PMC article. Review.
Cited by
-
Decreased age-related cardiac dysfunction, myocardial nitrative stress, inflammatory gene expression, and apoptosis in mice lacking fatty acid amide hydrolase.Am J Physiol Heart Circ Physiol. 2007 Aug;293(2):H909-18. doi: 10.1152/ajpheart.00373.2007. Epub 2007 Apr 13. Am J Physiol Heart Circ Physiol. 2007. PMID: 17434980 Free PMC article.
-
The Biological Functions and Regulatory Mechanisms of Fatty Acid Binding Protein 5 in Various Diseases.Front Cell Dev Biol. 2022 Apr 4;10:857919. doi: 10.3389/fcell.2022.857919. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35445019 Free PMC article. Review.
-
Cannabinoid receptors in acute and chronic complications of atherosclerosis.Br J Pharmacol. 2008 Jan;153(2):290-8. doi: 10.1038/sj.bjp.0707517. Epub 2007 Oct 29. Br J Pharmacol. 2008. PMID: 17965744 Free PMC article. Review.
-
Activation of the endocannabinoid system mediates cardiac hypertrophy induced by rosiglitazone.Acta Pharmacol Sin. 2022 Sep;43(9):2302-2312. doi: 10.1038/s41401-022-00858-x. Epub 2022 Feb 21. Acta Pharmacol Sin. 2022. PMID: 35190698 Free PMC article.
-
Cannabinoids as therapeutic agents in cardiovascular disease: a tale of passions and illusions.Br J Pharmacol. 2007 Jun;151(4):427-40. doi: 10.1038/sj.bjp.0707261. Epub 2007 Apr 23. Br J Pharmacol. 2007. PMID: 17450170 Free PMC article.
References
-
- Boger DL, Sato H, Lerner AE, Hedrick MP, Fecik RA, Miyauchi H, Wilkie GD, Austin BJ, Patricelli MP, Cravatt BF. Exceptionally potent inhibitors of fatty acid amide hydrolase: the enzyme responsible for degradation of endogenous oleamide and anandamide. Proc Natl Acad Sci USA. 2000;97:5044 –5049. - PMC - PubMed
-
- Bonz A, Laser M, Kullmer S, Kniesch S, Babin-Ebell J, Popp V, Ertl G, Wagner JA. Cannabinoids acting on CB1 receptors decrease contractile performance in human atrial muscle. J Cardiovasc Pharmacol. 2003;41:657– 664. - PubMed
-
- Burstein SH, Rossetti RG, Yagen B, Zurier RB. Oxidative metabolism of anandamide. Prostaglandins Other Lipid Mediat. 2000;61:29 – 41. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

